181816-48-8
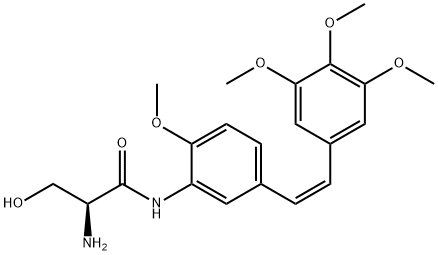 181816-48-8 結(jié)構(gòu)式
181816-48-8 結(jié)構(gòu)式
基本信息
(2S)-2-氨基-3-羥基-N-[2-甲氧基-5-[(1Z)-2-(3,4,5-三甲氧基苯基)乙烯基]苯基]丙酰胺
AVE-8062
Ombrabuli
ombrabulin
RPR-258062A.
CS-39-L-Ser.HCl
OMbrabulin(AVE-8062)
AVE8062
AC-7700
CS-39-L-SER.HCL
AVE-8062A
AVE-8062
RPR-258062A.
(S,Z)-2-Amino-3-hydroxy-N-(2-methoxy-5-(3,4,5-trimethoxystyryl)phenyl)propanamide
AVE8062
AVE8062A
AC7700
AVE-8062
AVE-8062A
AC-7700
AVE 8062
AVE 8062A
AC 7700
物理化學(xué)性質(zhì)
常見問題列表
tubulin
The effect of Ombrabulin (AC-7700) on endothelial or tumor cell viability is examined using the MTT assay. The IC 50 of Ombrabulin for the mouse mesenteric endothelial cells (MMEC) is 10 nM and ranges between 7 and 20 nM for the tumor cell lines (HeyA8, SKOV3ip1, and HeyA8-MDR). Comparative analysis of the nonlinear least-squares regression of the dose-response curves for each agent alone and combination Ombrabulin (AC-7700)/Docetaxel show a significantly lower IC 50 than either agent alone (P<0.005, all cell lines). The cytotoxicity of Docetaxel is 2- to 4-fold greater in combination with Ombrabulin (AC-7700) for the endothelial and tumor cells compared with Docetaxel alone.
Before performing therapy experiments, the tolerability of various doses of Ombrabulin (AC-7700) ranging from 10 to 100 mg/kg is tested given twice weekly via i.v., i.p., or s.c. routes in nude mice (n=3 per group). The i.v. and s.c. routes are not pursued further due to problems with skin or tail vein necrosis. The i.p. route is well tolerated with doses up to 100 mg/kg. Next, preliminary experiments are done to determine the lowest dose for in vivo therapeutic efficacy. Starting 7 days after tumor cell injection, nude mice (n=5 per group) bearing HeyA8 ovarian cancer cells are treated with either vehicle or Ombrabulin 10, 30, 50, and 100 mg/kg twice weekly i.p. for 3 weeks. There is 65% reduction in tumor weight in the 30 mg/kg group compared with the vehicle control group (P<0.02). The 10 mg/kg dose is not effective. The antitumor effects at doses >30 mg/kg are not significantly better; therefore, the 30 mg/kg dose is selected for subsequent therapy experiments.