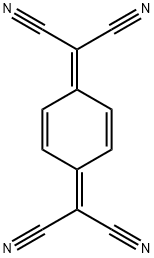
2,2'-(2,5-Cyclohexadien-1,4-diyli-den)bismalononitril
|
|
2,2'-(2,5-Cyclohexadien-1,4-diyli-den)bismalononitril Eigenschaften
- Schmelzpunkt:
- 287-289 °C (dec.) (lit.)
- Siedepunkt:
- 332.67°C (rough estimate)
- Dichte
- 1.3596 (rough estimate)
- Brechungsindex
- 1.5000 (estimate)
- storage temp.
- Sealed in dry,Room Temperature
- Aggregatzustand
- Crystalline Powder
- Farbe
- Orange-brown to green
- Wasserl?slichkeit
- insoluble
- Halbleitereigenschaften
- N-type (mobility=10-5cm2/V·s)
- maximale Wellenl?nge (λmax)
- 395nm(CH3CN)(lit.)
- BRN
- 611604
- Stabilit?t:
- Stable. Incompatible with strong acids, strong bases, strong reducing agents, strong oxidizing agents.
- InChI
- InChI=1S/C12H4N4/c13-5-11(6-14)9-1-2-10(4-3-9)12(7-15)8-16/h1-4H
- InChIKey
- PCCVSPMFGIFTHU-UHFFFAOYSA-N
- SMILES
- C1(=C(C#N)C#N)C=CC(=C(C#N)C#N)C=C1 |c:8,t:0|
- CAS Datenbank
- 1518-16-7(CAS DataBase Reference)
- NIST chemische Informationen
- Propanedinitrile, 2,2'-(2,5-cyclohexadiene-1,4-diylidene)bis-(1518-16-7)
- EPA chemische Informationen
- Propanedinitrile, 2,2'-(2,5-cyclohexadiene-1,4-diylidene)bis- (1518-16-7)
Sicherheit
| Kennzeichnung gef?hrlicher | Xn,Xi,T | ||
|---|---|---|---|
| R-S?tze: | 22-20/21/22-23/24/25 | ||
| S-S?tze: | 22-24/25-36/37-26-36-45 | ||
| RIDADR | 3276 | ||
| WGK Germany | 3 | ||
| RTECS-Nr. | GU4850000 | ||
| Hazard Note | Irritant | ||
| TSCA | Yes | ||
| HazardClass | 6.1 | ||
| PackingGroup | II | ||
| HS Code | 29269095 |
2,2'-(2,5-Cyclohexadien-1,4-diyli-den)bismalononitril Chemische Eigenschaften,Einsatz,Produktion Methoden
Beschreibung
7,7,8,8-tetracyanoquinodimethane (TCNQ), with a LUMO at 4.5 eV, is known for the charge-transfer salts formed by its radical anion TCNQ in?photovoltaic, light-emitting diodes, and organic field-effect transistor?devices. TCNQ and its derivatives?have been used as dopants, leading to an increase in hole mobility or to the lowering of injection?barriers. One classic example of such is the treatment of tetrathiafulvene (TTF), an electron donor with TCNQ. TFF and TCNQ form an ion pair, the TTF-TCNQ complex. This process of doping leads to the crystallisation of the ion pair into a one-dimensionally stacked polymer. This polymer consists of segregated stacks of cations and anions of the donors and the acceptors, respectively. The complex crystal is an organic semiconductor that exhibits metallic electric conductivity [1, 2].R-S?tze Betriebsanweisung:
R22:Gesundheitssch?dlich beim Verschlucken.R20/21/22:Gesundheitssch?dlich beim Einatmen,Verschlucken und Berührung mit der Haut.
S-S?tze Betriebsanweisung:
S22:Staub nicht einatmen.S24/25:Berührung mit den Augen und der Haut vermeiden.
S36/37:Bei der Arbeit geeignete Schutzhandschuhe und Schutzkleidung tragen.
S26:Bei Berührung mit den Augen sofort gründlich mit Wasser abspülen und Arzt konsultieren.
S36:DE: Bei der Arbeit geeignete Schutzkleidung tragen.
Chemische Eigenschaften
orange to green crystalline powderHistory
The first report on the electrical conductivity in an organic solid appeared in 1954, namely, a perylene—bromine complex with a room-temperature conductivity of 0.1 S cm?1. In 1960, the organic acceptor Tetracyanoquinodimethane (TCNQ; 7,7,8,8-Tetracyanoquinodimethane) was synthesized, as well as a great number of its conducting charge-transfer complexes and radical ion salts. In the 1970s, the organic donor TTF led to the first organic metal TTF-TCNQ. Its room-temperature conductivity (500 S cm?1) increases with a decrease in the temperature to the value of 6000 S cm?1 at 60 K, where a metal-insulator transition occurs[1].Verwenden
7,7,8,8-Tetracyanoquinodimethane is an electron-acceptor molecule used to form charge-transfer superconductors. It is an effective catalyst used for the ?-chlorination of carboxylic acids using chlorosulfonic acid; the presence of TCNQ suppresses competing free-radical chlorination.Definition
ChEBI: A quinodimethane that is p-quinodimethane in which the methylidene hydrogens are replaced by cyano groups.Allgemeine Beschreibung
7,7,8,8-Tetracyanoquinodimethane (TNCQ) is a strong electron acceptor as it has four cyano groups and π-conjugation bonds that form charge transferring chains and ion radical salts which are mainly used as p-dopants for the fabrication of a variety of semiconductor applications.2,2'-(2,5-Cyclohexadien-1,4-diyli-den)bismalononitril Upstream-Materialien And Downstream Produkte
Upstream-Materialien
Downstream Produkte
2,2'-(2,5-Cyclohexadien-1,4-diyli-den)bismalononitril Anbieter Lieferant Produzent Hersteller Vertrieb H?ndler.
Global( 223)Lieferanten
| Firmenname | Telefon | Land | Produktkatalog | Edge Rate | |
|---|---|---|---|---|---|
| Hubei Lidu New Material Technology Co., Ltd | +undefined+86-19307248857 |
liduxcl@163.com | China | 988 | 58 |
| Zibo Hangyu Biotechnology Development Co., Ltd | +86-0533-2185556 +8617865335152 |
Mandy@hangyubiotech.com | China | 10986 | 58 |
| Speranza Chemical Co., Ltd. | +86-86-075521030354 +8618688942810 |
sophieliu@speranzachem.com | China | 802 | 55 |
| Jilin Chinese Academy of Sciences - Yanshen Technology Co., Ltd. | 0431-80514535 13634302652 |
Extension@chemextension.com | CHINA | 967 | 58 |
| CONIER CHEM AND PHARMA LIMITED | +8618523575427 |
sales@conier.com | China | 49374 | 58 |
| career henan chemical co | +86-0371-86658258 +8613203830695 |
factory@coreychem.com | China | 29810 | 58 |
| Richest Group Ltd | 18017061086 |
oled@richest-group.com | CHINA | 5600 | 58 |
| Zhengzhou Alfa Chemical Co.,Ltd | +8618530059196 |
sale04@alfachem.cn | China | 11849 | 58 |
| Kindchem(Nanjing)Co.,Ltd | +86-025-025-85281586 +8618651653755 |
sales@kindchem.cn | China | 1226 | 58 |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418671 +8618949823763 |
sales@tnjchem.com | China | 34563 | 58 |
1518-16-7(2,2'-(2,5-Cyclohexadien-1,4-diyli-den)bismalononitril)Verwandte Suche:
4-Cyanbenzoylchlorid
4,4'-Diamino-diphenyl-methan
Diphenylmethan
Dibrommethan
Methyl-4-cyanbenzoat
Diiodmethan
Methylenedithiocyanate
2,3-Dimethoxy-5-methyl-p-benzochinon
Malons?uredinitril
Methyl-p-benzochinon
p-Benzochinon
2,2'-(2,5-Cyclohexadien-1,4-diyli-den)bismalononitril
- TIMTEC-BB SBB000435
- 2,2'-(2,5-cyclohexadiene-1,4-diylidene)bismalononitrile
- 2,5-Cyclohexadiene-delta(sup1alpha:4alpha)dimalononitrile
- 7,7,8,8-Tetracyano-1,4-chinodimethan
- 7,7,8,8-Tetracyano-1,4-quinodimethan
- 7,7,8,8-Tetracyanodimethan
- 7,7,8,8-Tetracyano-p-quinodimethane
- 7,7,8,8-tetracyanoquinoedimethane
- 7,7,8,8-tetracyanoquinonedimethane
- 7,7,8,8-Tetracyanoquino-Dimeth
- 7,7,8,8-Tetracyanoquinodimethane,98%TCNQ
- Propanedinitrile, 2,2-(2,5-cyclohexadiene-1,4-diylidene)bis-
- TCNQ/7,7'',8,8''-TETRACYANOQUINO-DIMETHANE
- 7,7,8,8-TETRACYANOQUINODIMETHANE TCNQ
- TCNQ/TETRACYANO-QUINODIMETHA
- Tetracyanoquinodimethane: (Ultima Gold AB)
- (2,5-Cyclohexadiene-1,4-diylidene)-dimalononitrile, TCNQ
- 2-[4-(Dicyanomethylidene)-1-cyclohexa-2,5-dienylidene]propanedinitrile
- 7,7,8,8-Tetracyanoquinodimethane,98%
- (2,5-Cyclohexadiene-1,4-diylidene)
- 7,7,8,8-Tetracyanoqu
- 2,2'-(Cyclohexa-2,5-diene-1,4-diylidene)dipropane-1,3-dinitrile, TCNQ
- 7,7,8,8-TetracyanoquinodiMethane, 98% 10GR
- 1,4-Bis(dicyanomethylene)cyclohexadiene TCNQ
- 2,2'-(Cyclohexa-2,5-diene-1,4-diylidene)diMalononitrile
- cyclohexa-2,5-diene-1,4-diylidene-bis-malononitrile
- Propanedinitrile,2,2’-(2,5-cyclohexadien-1,4-diylidene)bis-
- Quinodimethan, tetracyano-
- tetracyano-quinodimetha
- 1,4-BIS(DICYANOMETHYLENE)-2,5-CYCLOHEXADIENE
- 1,4-BIS(DICYANOMETHYLENE)CYCLOHEXADIENE
- 2,2'-(2,5-cyclohexadiene-1,4-diylidene)bispropanedinitrile
- 2,5-CYCLOHEXADIENE-1,4-DIYLIDENE-A,A'-DIMALONONITRILE
- (2,5-CYCLOHEXADIENE-1,4-DIYLIDENE)DIMALONONITRILE
- (2,5-CYCLOHEXADIENE-1,4-DIYLIDENE)MALONONITRILE
- 2,5-CYCLOHEXADIENE-DELTA1,ALPHA4,ALPHA'-DIMALONONITRILE
- TETRACYANO P-QUINODIMETHANE
- TETRACYANODIMETHANOQUINONE
- 7,7,8,8-TETRACYANO-1,4-QUINODIMETHANE
- 7,7,8,8-TETRACYANOQUINODIMETHANE
- 7,7,8,8-TetracyanoquinodiMethane (purified by subliMation)
- 7,7,8,8-TetracyanoquinodiMethane 98%
- 2,5-Cyclohexadiene-1,4-diylidene-alpha,alpha'-dimalononitrile
- 2,5-cyclohexadiene-delta(sup1,alpha:4,alpha)-dimalononitrile
- 2-[4-(dicyanomethylidene)cyclohexa-2,5-dien-1-ylidene]propanedinitrile
- 7,7,8,8-Tetracyanoquinonedimethane for synthesis
- 2,2’-(2,5-cyclohexadiene-1,4-diylidene)bis-propanedinitril
- cyclohexa-1,2,4,5-tetraene
- 7,7,8,8-Tetracyanoquinodimethane>
- 7,7,8,8-Tetracyanoquinodimethane, 98%, for synthesis
- TCNQ 7,7,8,8-Tetracyanoquinodimethan
- Tetracyano p-Quinone Dimethane
- Tetracyanoquinodimethan
- TCNQ
- TETRACYANOQUINODIMETHANE
- etracyanoquinodimethane
- 7,7',8,8'-tetracyanoquinodimethane
- 2,2'-(Cyclohexa-2,5-diene-1,4-diylidene)dimalononitrile