| Identification | More | [Name]
Potassium phosphate | [CAS]
7778-53-2 | [Synonyms]
BUFFER SOLUTION
POTASSIUM O-PHOSPHATE
POTASSIUM PHOSPHATE
POTASSIUM PHOSPHATE TRIBASIC
TKP
TRI-POTASSIUM ORTHOPHOSPHATE
TRIPOTASSIUM PHOSPHATE
Phosphoricacid,tripotassiumsalt
PotassiumPhosphateTribasicK3PO4
PotassiumPhosphateTribasicFcc
PotassiumPhosphateTribasicAcs
Tripotassiumphosphate,97%
Potassium phosphate tribasic, pure, 97%
Potassiumphosphate,min.97%
potassium phosphate, anhydrous
POTASSIUMPHOSPHATETRIBASIC,ANHYDROUS,REAGENT
POTASSIUM PHOSPHATE Dibasic Trihydrate EMD
TRIPOTASSIUMMONOPHOSPHATE
Tripotassium Orthphosphate
Potassium phosphate | [EINECS(EC#)]
231-907-1 | [Molecular Formula]
K3O4P | [MDL Number]
MFCD00036295 | [Molecular Weight]
212.27 | [MOL File]
7778-53-2.mol |
| Chemical Properties | Back Directory | [Appearance]
white powder or crystals | [Melting point ]
1340 °C
| [density ]
2.564 g/mL at 25 °C(lit.)
| [vapor pressure ]
0Pa at 20℃ | [storage temp. ]
room temp | [solubility ]
water: soluble100mg/mL, clear, colorless | [form ]
Solid | [color ]
White | [Specific Gravity]
2.564 | [Odor]
at 100.00?%. odorless | [PH]
11.9 (25°C, 10g/L) | [Stability:]
Stable. Incompatible with strong oxidizing agents, moisture. | [Water Solubility ]
50.8 g/100 mL (25 ºC) | [Sensitive ]
Hygroscopic | [Merck ]
14,7660 | [BCS Class]
1 | [CAS DataBase Reference]
7778-53-2(CAS DataBase Reference) | [EPA Substance Registry System]
7778-53-2(EPA Substance) |
| Safety Data | Back Directory | [Hazard Codes ]
C,Xi | [Risk Statements ]
R38:Irritating to the skin.
R41:Risk of serious damage to eyes.
R34:Causes burns. | [Safety Statements ]
S26:In case of contact with eyes, rinse immediately with plenty of water and seek medical advice .
S39:Wear eye/face protection .
S45:In case of accident or if you feel unwell, seek medical advice immediately (show label where possible) .
S36/37/39:Wear suitable protective clothing, gloves and eye/face protection . | [RIDADR ]
UN 1805 8/PG 3
| [WGK Germany ]
2
| [RTECS ]
TC8450000
| [F ]
8-10 | [TSCA ]
Yes | [HS Code ]
28352400 |
| Questions And Answer | Back Directory | [Description]
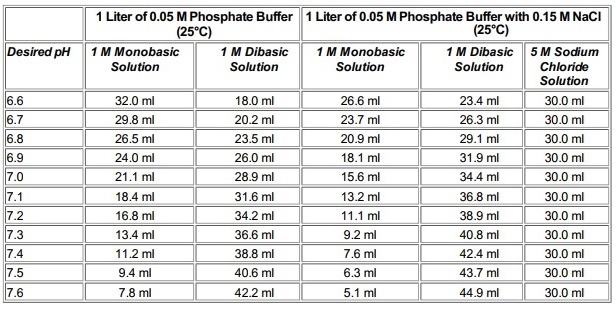
Potassium phosphate is a reagent with high buffering capacity, It serves as a buffering agent to regulate pH, Typically used as a component for a wide variety of media used in the culture of microorganisms, as a component in phosphate buffered saline (PBS). It occurs in several forms: monobasic, dibasic, and tribasic (K3PO4). Most pH neutral potassium phosphate buffer solutions consist of mixtures of the monobasic and dibasic forms to varying degrees, depending on the desired pH. Gomori buffers, the most commonly used phosphate buffers, consist of a mixture of monobasic dihydrogen phosphate and dibasic monohydrogen phosphate. By varying the amount of each salt, a range of buffers can be prepared that buffer well between pH 5.8 and pH 8.0 (please see the tables below). Phosphates have a very high buffering capacity and are highly soluble in water.
Potassium phosphate is used as a fertilizer and food additive. It is also used in optical modulators and for non-linear optics such as second-harmonic generation (SHG). It is involved in the phase-transfer catalyzed preparation of acetylenes from the corresponding vicinal dibromo compounds through double dehydrobromination. It is also useful for the oxidative coupling of thiols to disulfide.
| [Uses]
- Potassium phosphate is an acidity regulator and chelating agent (used to bind metal ions). It prevents desiccation and is used as an acid stabiliser in powders as well as to prevent formation of clumps. It increases the activity of antioxidants.
- In the food industry (Food additives), it is used as emulsifier, potassium fortifier, flavoring agent, meat binder. Potassium phosphate helps stabilize, thicken and regulate the acidity and moisture in foods. According to the provision of the FAO(1984), its application and limited amount is as below: instant broth; total phosphate salt: 1000 mg/kg (calculated based on P2O5); cooked pork forelegs, ham, cooked minced meat 3g/kg (single usage amount or combination amount with other phosphate, calculated on P2O5); low concentrated milk powder, condensed milk, dilute cream: 2 g/kg (single usage amount); combination amount with other stabilizer: 3 (calculated on anhydrous substance); milk powder, processed cheese: 5g/kg; processed cheese, the total amount of phosphate 9g/kg (calculated as phosphorus); cold drink: 2g/kg (single usage amount or combination amount with other phosphate, calculated on P2O5).
- It can be used for the preparation of pasta products with alkaline water.It can be used for the manufacture of liquid soap, high-quality paper, refined gasoline, high-quality paper, being the phosphorus and potassium fertilizers. It can be used as boiler water softener. Also used in medicine.Potassium phosphate is sometimes used as a medicine. It can have a diuretic effect and act as a laxative when used in medicinal amounts. However, phosphorus can interact with some medications, including corticosteroids and potassium-sparing diuretics, increasing the risk for adverse effects; only use it when advised to do so by a doctor.
- It can be used as analytical reagent, buffer and softener.
| [Toxicity]
ADI 0 to 70 (calculate based on the total phosphate as phosphorus; FAO/WHO, 2001).
ADI 0 to 70 mg/kg (calculated as phosphorus, FAO/WHO, 1985). It is generally recognized as being safe (US Food and Drug Administration, 1985). According to the provision of the FAO(1984); instant broth; total phosphate salt: 1000 mg/kg (calculated based on P2O5); cooked pork forelegs, ham, cooked minced meat 3g/kg; low concentrated milk powder, condensed milk, dilute cream: 2 g/kg; milk powder, processed cheese: 5g/kg; processed cheese, the total amount of phosphate 9g/kg (calculated as phosphorus); cold drink: 2g/kg. | [Side effects]
Phosphates are normal essential salts for the body. In view of the need to avoid calcium deficiency, their use is limited, as they bind up calcium readily. They have no side effects.
|
| Hazard Information | Back Directory | [Chemical Properties]
white powder or crystals | [Physical properties]
Colorless orthorhombic crystals; deliquescent; density 2.564 g/cm3at 17°C;melts at 1,380°C; very soluble in water, 90 g/100mL at 20°c; aqueous solutions strongly alkaline; insoluble in alcohol. | [Definition]
ChEBI: Tripotassium phosphate is an inorganic potassium salt that is the tripotassium salt of phosphoric acid. It is an inorganic phosphate salt and an inorganic potassium salt. | [Preparation]
The tribasic salt is produced by complete neutralization of phosphoric acid with potassium hydroxide, followed by evaporation and crystallization:H3PO4+ 3KOH →K3PO4+ 3H2O. | [General Description]
Potassium phosphate tribasic exhibits high catalytic property in trans esterification reactions. It has bactericidal activity. | [Flammability and Explosibility]
Notclassified |
|
|