| Identification | More | [Name]
Isophosphamide | [CAS]
3778-73-2 | [Synonyms]
3-(2-chloroethyl)-2-[(2-chloroethyl)amino]perhydro-2h-1,3,2-oxazaphosphorineoxide
3-(2-chloroethyl)-2-[(2-chloroethyl)amino]tetrahydro-2h-1,3,2-oxazaphosphorine 2-oxide
A-4942
ASTA Z-4942
IF
ifosfamid
IFOSFAMIDE
ISOPHOSPHAMIDE
N,3-BIS(2-CHLOROETHYL)TETRAHYDRO-2H-1,3,2-OXAZAPHOSPHORIN-2-AMINE 2-OXIDE
N,3-BIS(2-CHLOROETHYL)TETRAHYDRO-2H-1,3,2-OXAZAPHOSPHORINH-2-AMINE 2-OXIDE
NSC-1097
2,3-(n,n(sup1)-bis(2-chloroethyl)diamido)-1,3,2-oxazaphosphoridinoxyd
2-oxazaphosphorine,3-(2-chloroethyl)-2-((2-chloroethyl)amino)tetrahydro-3
3-(2-chloroethyl)-2-((2-chloroethyl)amino)perhydro-2h-1,3,2-oxazaphosphorine
3-(2-chloroethyl)-2-((2-chloroethyl)amino)tetrahydro-2h-1,3,2-oxazaphosphori
3,2-oxazaphosphorin-2-amine,n,3-bis(2-chloroethyl)tetrahydro-2h-2-oxide
3,2-oxazaphosphorine,3-(2-chloroethyl)-2-((2-chloroethyl)amino)tetrahydro-2-oxide
cyfos
holoxan
holoxan1000 | [EINECS(EC#)]
223-237-3 | [Molecular Formula]
C7H15Cl2N2O2P | [MDL Number]
MFCD00057374 | [Molecular Weight]
261.09 | [MOL File]
3778-73-2.mol |
| Chemical Properties | Back Directory | [Appearance]
Crystalline Solid | [Melting point ]
48°C | [Boiling point ]
336.1±52.0 °C(Predicted) | [density ]
1.33±0.1 g/cm3(Predicted) | [storage temp. ]
2-8°C | [solubility ]
DMF:50.0(Max Conc. mg/mL);191.51(Max Conc. mM)
DMSO:44.0(Max Conc. mg/mL);168.53(Max Conc. mM)
Ethanol:51.0(Max Conc. mg/mL);195.34(Max Conc. mM)
PBS (pH 7.2):10.0(Max Conc. mg/mL);38.3(Max Conc. mM)
Water:52.0(Max Conc. mg/mL);199.17(Max Conc. mM) | [form ]
neat | [pka]
1.44±0.20(Predicted) | [color ]
White to off-white | [Usage]
A cytostatic agent, related structurally to cyclophosphamide | [CAS DataBase Reference]
3778-73-2(CAS DataBase Reference) | [IARC]
3 (Vol. 26, Sup 7) 1987 | [EPA Substance Registry System]
Isophosphamide (3778-73-2) |
| Safety Data | Back Directory | [Hazard Codes ]
T | [Risk Statements ]
R25:Toxic if swallowed.
R36:Irritating to the eyes. | [Safety Statements ]
S26:In case of contact with eyes, rinse immediately with plenty of water and seek medical advice .
S45:In case of accident or if you feel unwell, seek medical advice immediately (show label where possible) . | [RIDADR ]
3249 | [WGK Germany ]
3 | [RTECS ]
RP6050000 | [HazardClass ]
6.1(b) | [PackingGroup ]
III | [HS Code ]
29349900 | [Hazardous Substances Data]
3778-73-2(Hazardous Substances Data) | [Toxicity]
LD50 in rats (mg/kg): 160 i.p. (Arnold, 1973); also reported as 150 i.p. (Brock) |
| Questions And Answer | Back Directory | [History]
H. Arnold at ASTA Inc. developed a new drug ifosfamide in 1967 by modifying cyclophosphamide. This drug, which is a structural isomer of cyclophosphamide, was not only effective for leukemia treatment but also showed a superior effi cacy compared to cyclophosphamide in solid tumor such as testicular cancer and sarcoma. Therefore, it obtained FDA approval as a therapeutic agent for testicular cancer and sarcoma in 1988. In 1981, while searching for approaches to lower the toxicity of ifosfamide, N. Brock et al. discovered that sodium-2-mercaptoethane sulfonate (mesna), which contains a thiol group, can convert acrolein into a non-toxic compound and thereby effectively prevent bladder toxicity (hemorrhagic cystitis). Since then, mesna has been used until today to clinically prevent the side effects of ifosfamide.
| [Cyclophosphamide isomer]
Ifosfamide is a cyclophosphamide isomer, appearing as white crystalline or crystalline powder at room temperature. The difference with cyclophosphamide is the difference of the position of a chloroethyl. Itself is a latent drug and can take effect only after entering into the body to be activated in the liver. It is used for the treatment of bone and soft tissue sarcoma, non-small cell lung cancer, breast cancer, head and neck cancer, cervical cancer, esophageal cancer, the effect is better than cyclophosphamide.
Cyclophosphamide is the most commonly used alkylating agent antitumor drugs. After entering into the body, it is catalyzed by the liver microsomal enzyme to be decomposed for release of strong alkylation chloroethyl phosphate amide (or phosphamide nitrogen mustard) with cytotoxic effect on tumor cells. In addition, this product also has a significant immune function.
It is clinically used for the treatment of malignant lymphoma, multiple myeloma, leukemia, breast cancer, ovarian cancer, cervical cancer, prostate cancer, colon cancer, bronchial cancer and lung cancer with a certain effect. It can also be used for the treatment of rheumatoid arthritis, nephrotic syndrome in children and autoimmune diseases. | [Instructions for ifosfamide (for injection)]
Properties: This product is white crystalline or crystalline powder.
Drug categories: alkylating agents antitumor drugs
Pharmacology and Toxicology: This product has no anti-cancer activity in vitro, entering into the body to be hydrolyzed by the phosphorylase or phosphatase in the liver or tumor, becoming activating form of phosphoramide nitrogen mustard. Its mechanism of action can have cross-linking with DNA, causing inhibition of DNA synthesis and interfering with the function of RNA, belonging to cell cycle non-specific drugs. This product has broad anti-tumor spectrum with inhibitory effect on a variety of tumors.
Pharmacokinetics: it is subject to an intravenous infusion 3.8~5.0g/m2 according to the surface area with the plasma concentration being biphasic and terminal half-life of 15 hours. It is subject to an intravenous infusion of 1.6~2.4g/m2 based on the surface area with the plasma concentration exhibiting single Phase with a half-life of 7 hours. It can be degraded by the liver with only a few amounts of its active metabolites penetrating through the blood-brain barrier. Drugs discharged by the kidneys accounts for 70% to 80%; upon intravenous injection of 5.0g/m2 once according to the surface area, 61% is discharged in the form of prototype; upon intravenous injection of 1.2~2.4g/m2 once according to the surface area, only 12% to 18% is discharged in the form of prototype .
Indications: for testicular cancer, ovarian cancer, breast cancer, sarcoma, malignant lymphoma and lung cancer.
Dosage: single drug treatment: apply intravenous injection based on a body surface area of 1.2~2.5g/m2 each time with 5 days as a course of treatment. Combination treatment: apply intravenous injection at 1.2~2.0g/m2 each time based on body surface area with continuous 5 days as a course of treatment. Each course has a 3 to 4 weeks gap with 500 to 600 mg/m2.
Adverse reactions: (1) bone marrow suppression: Leukopenia is more common than thrombocytopenia with the lowest value occurring in 1 to 2 weeks after treatment and will usually recover in 2 to 3 weeks after recovery. It has effect on the liver function. Gastrointestinal reactions: including loss of appetite, nausea and vomiting. It will generally disappear after drug withdrawal in 1 to 3 days.
(2) Urinary tract reaction: can cause hemorrhagic cystitis, manifested as dysuria, urinary frequency and dysuria. This may occur within a few hours or weeks after administration, usually disappear within a few days after stopping the drug.
(3) Central nervous system toxicity: dose-related, usually manifested as anxiety, confusion, hallucinations and lack of strengthen. In rare cases, there will be syncope, epileptic seizures and even coma.
(4) Rarely: transient asymptomatic liver and kidney dysfunction; upon high dose of medication, there may be metabolic acidosis due to renal toxicity. It is rarely seen of heart and lung toxicity.
(5) Other reactions include hair loss, nausea and vomiting. It can occur of phlebitis in the injection site.
(6) Long-term medication can produce immune suppression, pituitary dysfunction, infertility and secondary tumors. | [Contraindications and Precautions]
Contraindications
Patients of severe bone marrow suppression being allergic to the products and pregnancy or lactating women should be disabled.
Precautions
(1) The metabolic product of this product has urinary tract irritation. During application, it should be encouraged of patients to drink more water with high-dose application should be hydration, diuretic while giving the protective agent of urinary tract, Mesna.
(2) Patients of hypoalbuminemia, liver and kidney dysfunction, bone marrow suppression and women of childbearing age should use with caution.
(3) This product is unstable aqueous solution, shall be freshly prepared before use.
(4) During treatment, it should be regularly monitored of the white blood cells, platelets and carried out of liver and kidney function determination. Administration of pregnant women and lactating women has mutagenic, teratogenic effects, being able to cause fetal death or congenital malformations. Therefore, pregnant women should be disabled. This product can be discharged in the milk and should be stopped of breast-feeding in the beginning of the medication. | [Medicine interactions]
(1) Patients previously used cisplatin may get aggravated ifosfamide inhibition of bone marrow, neurotoxicity and renal toxicity.
(2) Simultaneous administration of anticoagulant drugs may lead to bleeding risk.
(3) Simultaneous administration of hypoglycemic agents can enhance hypoglycemic effect.
(4) Upon simultaneous administration with other cytotoxic drugs, it should be considered of appropriate reduction.
Storage: shading, closed, stored at below 30 ℃.
Packing: (1) 0.5g (2) 1.0g | [Uses]
1. Anti-cancer drugs;
2. For the treatment of malignant lymphoma, lung cancer, breast cancer, esophageal cancer, bone and soft tissue sarcoma, non-small cell lung cancer, head and neck cancer, cervical cancer | [The domestic market situation]
Because the ifosfamide products have much higher price than cyclophosphamide products, so only upon no return in the production of cyclophosphamide, the several major domestic companies of production of cyclophosphamide have changed production lines for production of ifosfamide.
Ifosfamide is cyclophosphamide derivatives, having in vitro activity, but administration in vivo can have a significant inhibitory effect on a variety of metastatic tumors, being able to treat bone and soft tissue sarcoma, non-small cell lung cancer, breast cancer, head and neck cancer, cervical cancer and esophageal cancer. In 1993, it is officially available in the country.
It is understood that the current market price of ifosfamide bulk drugs is more than 8000 yuan/kg. The preparation class drugs, after 18 times of price decreases, can decrease from the original 216 yuan down to 180 yuan each one. The corporate profit margin has decreased, but is still considerable compared to cyclophosphamide.
In the last year, in the rankings list of 42 anti-tumor drugs, ifosfamide ranked 13. In the 14 varieties of the world's leading sales of antineoplastic drugs of 2003, ifosfamide ranked 11 in a sale of 08 million US dollars. The market performance is still stable. According to the reflection of domestic enterprises, the annual demand of ifosfamide preparation is not large, so raw material supply and demand situation is relatively stable.
However, one of the largest manufacturers of anti-tumor drug, the Jiangsu Hengrui, after discontinuing the production line of cyclophosphamide, has also significantly expanded the production of ifosfamide. It is understood that at this year, a securities company's research report on the Hengrui has shown that, ifosfamide needle has obtained an increased production of 650,000 with the new output value of 78 million yuan. Ifosfamide and other anti-cancer drugs have been simultaneously listed important nurturing products. Therefore, it can be expected that, in the circumstances of downturn of cyclophosphamide market, companies have converted to ifosfamide with the latter's market potentially getting a larger growth.
The reason why the latter has been able to get the favor of enterprises is mainly due to the price. Although the production of ifosfamide cyclophosphamide than the process is much more complex and costly, but the market price of 180 yuan/branch can still make the enterprise more profitable, while the production of cyclophosphamide is a high cost with zero return. |
| Hazard Information | Back Directory | [Chemical Properties]
Crystalline Solid | [Originator]
Holoxan,Lucien,France,1976 | [Definition]
ChEBI: The simplest member of the class of ifosfamides that is 1,3,2-oxazaphosphinan-2-amine 2-oxide substituted by 2-chloroethyl groups on both the nitrogen atoms respectively. It is a nitrogen mustard alkylating agent used in the treatment of advanced breast c
ncer. | [Indications]
Ifosfamide (Ifex) is an analogue of cyclophosphamide
that requires metabolic activation to form 4-hydroxyifosfamide.
In general, the metabolism, serum half-life,
and excretion of ifosfamide are similar to those of cyclophosphamide.
Ifosfamide is active against a broad spectrum of tumors,
including germ cell cancers of the testis, lymphomas,
sarcomas, and carcinomas of the lung, breast,
and ovary. It is thought to be more active than cyclophosphamide
in germ cell cancers and sarcomas.
Ifosfamide is less myelosuppressive than cyclophosphamide
but is more toxic to the bladder. It also may
produce alopecia, nausea, vomiting, infertility, and second
tumors, particularly acute leukemias. Neurological
symptoms including confusion, somnolence, and hallucinations
have also been reported. It is recommended
that ifosfamide be coadministered with the thiol compound
mesna (Mesnex) to avoid hemorrhagic cystitis. | [Manufacturing Process]
127.6 g (1.1 mols) of N-(2-chloroethyl)-amine hydrochloride are suspended in
a solution of 218 g (1 mol) of N-(2-chloroethyl)-N,O-propylene phosphoric
acid ester amide monochloride in 600 cc of methylene dichloride, and 212 g of
triethylamine are added thereto dropwise with stirring. The reaction mixture is
heated to boiling by the reaction heat. After termination of the addition, the
reaction mixture is heated to boiling for another 2 hours. Thereafter, it is
cooled to room temperature and the precipitated triethylamine hydrochloride
is separated by filtration with suction. The filtrate is extracted with about 60cc
of dilute hydrochloric acid (pH 3), then twice with about 60 cc of water,
thereafter with about 60 cc of dilute soda lye and finally twice with about 60
cc of water. After drying over anhydrous sodium sulfate, methylene dichloride
is distilled off under normal pressure. The oily residue is dried in a vacuum
and thereafter extracted in a perforator with 500 cc of anhydrous ether. The
oily extract crystallizes upon inoculation and standing in an ice box. After
standing for several hours, the precipitate is filtered off, washed with a small
amount of cold ether and dried in a vacuum at room temperature. Yield: 185
g (71% of the theoretical). This material is also identified as 3-(2-
chloroethyl)-2-(2-chloroethylamino)-tetrahydro-2H-1,3,2-oxazaphosphorin-2-
oxide; generic name: ifosfamide. F.P.: 39°C to 41°C. | [Brand name]
Ifex (Bristol-Myers Squibb). | [Therapeutic Function]
Antineoplastic | [General Description]
Ifosfamide is available in 1- and 3-g vials for IV administrationas Food and Drug Administration (FDA)-approvedthird-line therapy in the treatment of testicular cancer. It has also been utilized (although not FDA approved) in the treatmentof a wide variety of cancers including Hodgkin’s andnon-Hodgkin’s lymphoma, soft tissue sarcoma, germ celltumors, small cell lung cancer, non–small cell lung cancer(NSCLC), cancers of the head and neck, bladder cancer, cervicalcancer, and Ewing sarcoma. Coadministration ofmesna is recommended. The mechanisms of resistance areidentical to those seen with cyclophosphamide. The drug iswidely distributed with a low extent of protein binding(20%). Metabolism primarily by CYP3A4/5 and CYP2B6 isrequired for activation of the compound. Theagent is administered as the racemic mixture as a result ofthe presence of the chiral phosphorus atom, and differentialmetabolism of the R- and S-isomers has been observed. Incontrast to cyclophosphamide, there is a greater amount ofdeactivation of the agent by N-dechloroethylation and subsequentlymore chloroacetaldehyde is produced, which mayresult in a greater amount of neurotoxicity and nephrotoxicitythan seen with cyclophosphamide. The N-dechloroethylatedmetabolites are the predominate species seen in theplasma. The parent and metabolites are eliminated inthe urine with an elimination half-life of 3 to 10 hours. Themajor components found in the urine are the dechlorethylatedmetabolites. Dose-limiting toxicities include myelosuppressionand bladder toxicity. Other adverse effectsinclude nausea, alopecia, amenorrhea, inappropriate secretionof antidiuretic hormone, as well as the production ofsecondary cancers. Neurotoxicity, which is associated withthe production of chloroacetaldehyde presents as confusion,seizure, weakness, and hallucination, and coma may occur. | [Biological Activity]
Ifosfamide is a nitrogen mustard compound th at is a structural isomer of cyclophosphamide. Ifosfamide is a prodrug th at must be transformed by cytochrome P450 to the biologically active component. It is used as an antineoplastic agent in cancer chemotherapybut ifosfamide is more likely to cause renal toxicity than cyclophosphamide. | [Clinical Use]
Ifosamide currently is used as “third-line” therapy against testicular cancer, although it also has shown activity in a number of solid tumors and hematologic cancers. | [Side effects]
Patients on ifosfamide (but not cyclophosphamide) commonly exhibit central nervous system (CNS) toxicity. In severe forms, CNS depression can progress to coma and death. | [Synthesis]
Ifosfamide, 3-(2-chloroethyl)-2-[(2-chloroethyl)amino]tetrahydro-2H-1,3,2-
oxazaphosphorin-2-oxide (30.2.1.17), which is viewed as an isomeric compound of
cyclophosphamide (30.2.1.15) and which is analogous in terms of action, is made by react�ing N-(2-chloroethyl)-N-(3-hydroxypropyl)amine with phosphorous oxychloride, giving
3-(2-chloroethyl)-2-chlorotetrahydro-2H-1,3,2-oxazaphosphorin-2-oxide (30.2.1.16),
which is reacted with N-(2-chloroethyl)amine, forming the desired ifosfamide (30.2.1.17).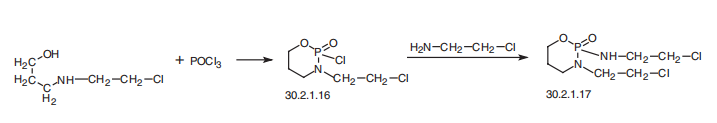
| [Veterinary Drugs and Treatments]
In small animals, ifosfamide may be of benefit as part of treatment
protocols for a variety of neoplasms. Treatment of lymphomas and
soft tissue sarcomas with ifosfamide in dogs and cats has been investigated
to some extent; some efficacy has been demonstrated.
In humans, ifosfamide is used in various treatment protocols for
testicular neoplasms, bone and soft tissue sarcomas, bladder cancer,
lung cancer, cervical cancer, ovarian cancer, and some types of
lymphomas. | [Drug interactions]
Potentially hazardous interactions with other drugs
Anticoagulants: possibly enhanced effect of
coumarins.
Antipsychotics: avoid concomitant use with
clozapine (increased risk of agranulocytosis).
| [Metabolism]
The pharmacokinetics of ifosfamide are reported to
exhibit considerable inter-individual variation. It is
a prodrug that is extensively metabolised, chiefly by
cytochrome P450 isoenzymes CYP3A4 and CYP2B6 in
the liver, to both active and inactive alkylating metabolites;
there is some evidence that metabolism is saturated
at very high doses. After repeated doses (fractionated
therapy) there is a decrease in the elimination half-life,
apparently due to auto-induction of metabolism. It is
excreted largely in urine, as unchanged drug (80%) and
metabolites. |
|
|