| Identification | More | [Name]
1,3-BUTADIENE | [CAS]
106-99-0 | [Synonyms]
1,3-BUTADIENE
BIETHYLENE
BIVINYL
BUDIENE
BUTADIENE
DIVINYL
ERYTHRENE
PYRROLYLENE
VINYLETHYLENE
(E)-CH2=CHCH=CH2
1-methylallene
a,g-Butadiene
alpha,gamma-Butadiene
alpha-butadiene
Buta-1,3-dieen
Buta-1,3-dien
Butadieen
Butadien
butadien(polish)
butadiene(non-specificname) | [EINECS(EC#)]
203-450-8 | [Molecular Formula]
C4H6 | [MDL Number]
MFCD00008659 | [Molecular Weight]
54.09 | [MOL File]
106-99-0.mol |
| Chemical Properties | Back Directory | [Description]
1,3-Butadiene is a simple conjugated diene. It is a colourless gas with a mild aromatic or
gasoline-like odour and incompatible with phenol, chlorine dioxide, copper, and crotonaldehyde.
The gas is heavier than air and may travel along the ground; distant ignition
is possible. It is an important industrial chemical used as a monomer in the production
of synthetic rubber. Most butadiene is polymerised to produce synthetic rubber. While
polybutadiene itself is a very soft, almost liquid, material, polymers prepared from
mixtures of butadiene with styrene or acrylonitrile, such as ABS, are both tough and
elastic. Styrene–butadiene rubber is the material most commonly used for the production
of automobile tyres. Smaller amounts of butadiene are used to make nylon via the intermediate adiponitrile, other synthetic rubber materials such as chloroprene, and the
solvent sulpholane. Butadiene is used in the industrial production of cyclododecatriene
via a trimerisation reaction. | [Appearance]
colourless gas, or pressurised liquid (odour TLV 1.6 ppm) | [Melting point ]
−109 °C(lit.)
| [Boiling point ]
−4.5 °C(lit.)
| [density ]
0.62 g/mL at 20 °C(lit.)
| [vapor density ]
1.9 (15 °C, vs air)
| [vapor pressure ]
1863 mm Hg ( 21 °C)
| [refractive index ]
1.4292 | [Fp ]
−105 °F
| [storage temp. ]
0-6°C | [solubility ]
water: soluble0.5g/L at 20°C | [form ]
Colorless gas | [color ]
Colorless to Almost colorless | [Stability:]
Stable. Extremely flammable. May form explosive mixtures with air. Incompatible with strong oxidizing agents, copper, copper alloys. May contain stabilizer. | [explosive limit]
12% | [Odor Threshold]
0.23ppm | [Water Solubility ]
735mg/L(25 ºC) | [FreezingPoint ]
-108.91℃ | [Merck ]
1509 | [BRN ]
605258 | [Henry's Law Constant]
(x 10-2 atm?m3/mol):
6.3 at 25 °C (Hine and Mookerjee, 1975) | [Exposure limits]
TLV-TWA 10 ppm (~22 mg/m3) (ACGIH),
1000 ppm (OSHA and NIOSH); IDLH
20,000 ppm (NIOSH); A2–Suspected Human
Carcinogen (ACGIH). | [InChIKey]
KAKZBPTYRLMSJV-UHFFFAOYSA-N | [LogP]
1.99 at 20℃ | [Uses]
Synthetic elastomers (styrene-butadiene,
polybutadiene, neoprene, nitriles), ABS resins,
chemical intermediate.
| [CAS DataBase Reference]
106-99-0(CAS DataBase Reference) | [IARC]
1 (Vol. Sup 7, 54, 71, 97, 100F) 2012 | [EPA Substance Registry System]
1,3-Butadiene (106-99-0) |
| Hazard Information | Back Directory | [Chemical Properties]
1,3-Butadiene is a simple conjugated diene. It is a colorless gas with a mild aromatic or
gasoline-like odor and incompatible with phenol, chlorine dioxide, copper, and crotonaldehyde. The gas is heavier than air and may travel along the ground; distant ignition is
possible. It is an important industrial chemical used as a monomer in the production of
synthetic rubber. Most butadiene is polymerized to produce synthetic rubber. While polybutadiene itself is a very soft, almost liquid material, polymers prepared from mixtures of
butadiene with styrene or acrylonitrile, such as ABS, are both tough and elastic. Styrenebutadiene rubber is the material most commonly used for the production of automobile
tires. Smaller amounts of butadiene are used to make nylon via the intermediate adiponitrile, other synthetic rubber materials such as chloroprene, and the solvent sulfolane.
Butadiene is used in the industrial production of cyclododecatriene via a trimerization
reaction. | [General Description]
Butadiene, inhibited is a colorless gas with an aromatic odor. 1,3-BUTADIENE(106-99-0) is shipped as a liquefied gas under its vapor pressure. Contact with the liquid can cause frostbite. 1,3-BUTADIENE(106-99-0) is easily ignited. Its vapors are heavier than air and a flame can flash back to the source of leak very easily. 1,3-BUTADIENE(106-99-0) can asphyxiate by the displacement of air. 1,3-BUTADIENE(106-99-0) must be shipped inhibited as butadiene is liable to polymerization. If polymerization occurs in the container, 1,3-BUTADIENE(106-99-0) may violently rupture. Under prolonged exposure to fire or intense heat the containers may rupture violently and rocket. 1,3-BUTADIENE(106-99-0) is used to make synthetic rubber and plastics, and to make other chemicals. | [Reactivity Profile]
A colorless gas, 1,3-BUTADIENE can react with oxidizing reagents. Upon long exposure to air 1,3-BUTADIENE forms explosive peroxides. They are sensitive to heat or shock; sudden polymerization may occur [Scott, D. A., Chem. Eng. News, 1940, 18, p.404]. Butadiene polyperoxides are insoluble in liquefied butadiene (m. p.-113° C, b. p.-2.6° C) and progressively separate leading to local concentration build up. Self-heating from a spontaneous decomposition will lead to explosion [Hendry, D. G. et al., Ind. Eng. Chem., 1968, 7, p. 136, 1145]. Explodes on contact with aluminum tetrahydroborate, potentially explosive reaction with chlorine dioxide (peroxide) and crotonaldehyde (above 180° C). Reaction with sodium nitrite forms a spontaneously flammable product [Sax, 9th ed., 1996, p. 539]. | [Air & Water Reactions]
Highly flammable. In contact with air, butadiene may form violently explosive peroxides, which can be exploded by mild heat or shock. Solid butadiene absorbs enough oxygen at sub atmospheric pressures to make 1,3-BUTADIENE explode violently when heated just above its melting point [Ind. Eng. Chem. 51:733 1959]. | [Hazard]
A confirmed carcinogen. Irritant in high
concentration. Highly flammable gas or liquid,
explosive limits in air 2–11%. May form explosive
peroxides on exposure to air. Must be kept inhibited
during storage and shipment. Inhibitors often used
are di-n-butylamine or phenyl-β-naphthylamine.
Storage is usually under pressure or in insulated
tanks <35F (<1.67C).
| [Health Hazard]
Slight anesthetic effect at high concentrations; causes ``frostbite'' from skin contact; slight irritation to eyes and nose at high concentrations. | [Health Hazard]
The symptoms of poisoning include distorted blurred vision, vertigo, general tiredness, decreased blood pressure, headache, and nausea. Exposures to very high concentrations of 1,3-butadiene are known to cause CNS depression, decreased pulse rate,
drowsiness, fatigue, vertigo, ataxia, unconsciousness, coma, respiratory paralysis, and
death. Several studies show butadiene exposure increases the risk of cardiovascular
diseases and cancer. There is a lack of human data on the effects butadiene. Animal
studies have shown breathing butadiene during pregnancy can increase the number
of birth defects. | [Fire Hazard]
Behavior in Fire: Vapors heavier than air and may travel a considerable distance to a source of ignition and flashback. Containers may explode in a fire due to polymerization. | [Physical properties]
Colorless gas with a mild, aromatic or gasoline-like odor. Experimentally determined detection
and recognition odor threshold concentrations were 1.0 mg/m3 (0.45 ppmv) and 2.4 mg/m3 (1.1
ppmv), respectively (Hellman and Small, 1974). | [Definition]
ChEBI: A butadiene with unsaturation at positions 1 and 3. | [Preparation]
Butadiene is obtained when n-butenes are dehydrogenated: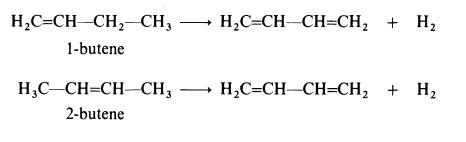
n-Butenes are mostly obtained from the catalytic cracking operations carried
out on various petroleum fractions; thermal cracking processes usually give
low yields of butenes. The dehydrogenation of n-butenes is carried out by
mixing the feed with steam (which lowers the partial pressures of the
reactants) and passing over a catalyst such as mixed calcium/nickel phos�phate stabilized with chromium oxide at about 650??C. | [Production Methods]
Except for a small amount of butadiene produced by the
oxydehydrogenation of n-butane, most of butadiene is produced
commercially as a by-product of ethylene production
during the steam cracking of hydrocarbon streams. It is
separated and purificated from other components by extractive
distillation, using acetonitrile and dimethylformamide as
solvents. | [Flammability and Explosibility]
Extremelyflammable | [Biochem/physiol Actions]
Environmental carcinogen. Induces cardiac hemangiosarcomas in mice. | [Materials Uses]
1,3-Butadiene is noncorrosive and may be used
with any common metals. Steel is recommended
for tanks and piping in butadiene service by
some authorities. If used with plastics, compatibility
must be confirmed. Welded rather than
threaded connections are similarly recommended
because 1,3-butadiene tends to leak
through even extremely small openings. If
threaded connections are used, Schedule 80
pipe should be used. Before being exposed to
1,3-butadiene that is not inhibited, iron surfaces
should be treated with an appropriate reducing
agent such as sodium nitrite because polymerization
is accelerated by oxygen (even if present
as in ferrous oxide), as well as by heat. | [Potential Exposure]
Tumorigen,Mutagen; Reproductive Effector; Human Data. 1,3-Butadiene is used chiefly as the principal monomer in themanufacture of many types of synthetic rubber and otherchemicals. Butadiene is finding increasing usage in the formation of rocket fuels, plastics, and resins. | [Physiological effects]
If inhaled in high concentrations, 1,3-butadiene
has an anesthetic or mild narcotic action, which
appears to vary with individuals. Inhalation of a
I percent concentration in air has been reported
to have had no effect on the respiration or blood
pressure of individuals. However, such exposure
may cause the pulse rate to quicken and
give a sensation of prickling and dryness in the
nose and mouth. Inhalation in higher concentrations
has brought on blurring of vision and nausea
in some persons. Inhalation in excessive
amounts leads to progressive anesthesia, irritation
of eyes, lungs, and nasal passages. Exposure
to a 25 percent concentration for 23 minutes
proved fatal in one instance. No cumulative
action on the blood, lungs, liver, or kidneys has
been reported. Because 1,3-butadiene liquid
evaporates rapidly, prolonged contact between
liquid butadiene and the skin causes freezing of
the tissue. Delayed skin bums may result if liquid
butadiene is allowed to remain trapped in
clothing or in shoes.
OSHA has concluded that there is strong evidence
that workplace exposure to 1,3-butadiene
poses an increased risk of death from cancers of
the Iymphokematopoietic system. ACGIH
has classified 1,3-butadiene as a "suspected
human carcinogen" . The National Toxicology
Program has classified 1,3-butadiene as
showing clear evidence of carcinogenicity. | [First aid]
If this chemical gets into the eyes, remove anycontact lenses at once and irrigate immediately for atleast 15 min, occasionally lifting upper and lower lids.Seek medical attention immediately. If this chemical contacts the skin, remove contaminated clothing and washimmediately with soap and water. Seek medical attentionimmediately. If this chemical has been inhaled, removefrom exposure, begin rescue breathing (using universal precautions, including resuscitation mask) if breathing hasstopped and CPR if heart action has stopped. Transferpromptly to a medical facility. When this chemical hasbeen swallowed, get medical attention. Give large quantities of water and induce vomiting. Do not make an unconscious person vomit. If frostbite has occurred, seek medicalattention immediately; do NOT rub the affected areas orflush them with water. In order to prevent further tissuedamage, do NOT attempt to remove frozen clothing fromfrostbitten areas. If frostbite has NOT occurred, immediately and thoroughly wash contaminated skin with soapand water. | [Carcinogenicity]
1,3-Butadiene is known to be a human carcinogen based on sufficient evidence of carcinogenicity from studies in humans, including epidemiological and mechanistic studies. 1,3-Butadiene was first listed in the Fifth Annual Report on Carcinogens in 1989 as reasonably anticipated to be a human carcinogen based on sufficient evidence of carcinogenicity from studies in experimental animals. The listing was revised to known to be a human carcinogen in the Ninth Report on Carcinogens in 2000. | [Source]
Schauer et al. (2001) measured organic compound emission rates for volatile organic compounds, gas-phase semi-volatile organic compounds, and particle-phase organic compounds
from the residential (fireplace) combustion of pine, oak, and eucalyptus. The gas-phase emission
rate of 1,3-butadiene was 177 mg/kg of pine burned. Emission rates of 1,3-butadiene were not
measured during the combustion of oak and eucalyptus. | [Environmental Fate]
Surface Water. The estimated volatilization half-life of 1,3-butadiene in a model river 1 m deep,
flowing 1 m/sec and a wind speed of 3 m/sec is 3.8 h (Lyman et al., 1982).
Photolytic. The following rate constants were reported for the reaction of 1,3-butadiene and OH
radicals in the atmosphere: 6.9 x 10-11 cm3/molecule·sec (Atkinson et al., 1979) and 6.7 x 10-11
cm3/molecule·sec (Sablji? and Güsten, 1990). Atkinson and Carter (1984) reported a rate constant
of 6.7–8.4 x 10-11 cm3/molecule·sec for the reaction of 1,3-butadiene and ozone in the atmosphere.
Photooxidation reaction rate constants of 2.13 x 10-13 and 7.50 x 10-18 cm3/molecule·sec were
reported for the reaction of 1,3-butadiene and NO3 (Benter and Schindler, 1988; Sablji? and
Güsten, 1990). The half-life in air for the reaction of 1,3-butadiene and NO3 radicals is 15 h
(Atkinson et al., 1984a).
Chemical/Physical. Will polymerize in the presence of oxygen if no inhibitor is present
(Hawley, 1981). | [storage]
1,3-Butadiene is stored in a cool and wellventilatedlocation separated from combustibleand oxidizing substances. Smallamounts of stabilizers, such as o-dihydroxybenzene,p-tert-butylcatechol, or aliphaticmercaptans, are added to prevent its polymerizationor peroxides formation. The cylindersare stored vertically and protected againstphysical damage. | [Shipping]
Butadiene, inhibited, requires a shipping label of“FLAMMABLE GAS.” This material falls in Hazard Class2.1 and does not have an assigned Packing Group.[19, 20] | [Toxicity evaluation]
Butadiene is a gas under normal environmental conditions
with limited water solubility (735 mg l-1 at 25°C). Butadiene
released to the atmosphere will remain there with very
small amounts being distributed to water and soil. In air,
butadiene will be removed by reaction with photochemically
produced hydroxyl radicals (5.6-h half-life), nitrate radicals
(15-h half-life), and ozone (1.5-day half-life). When released
to water, butadiene will be removed by volatilization to air
(Henry’s law constant of 7460 Pam3 mol-1), biodegradation
(aerobic half-life of 15 days), and reaction with singlet oxygen. Based on its estimated organic carbon partition
coefficient (Koc of 288), butadiene will not exhibit significant
adsorption to soil or suspended particulate matter; its
biodegradation half-life in soil is estimated to be 30 days.
Due to volatilization to air and degradation in soil, butadiene
is not expected to leach to groundwater. As butadiene is
readily metabolized, it is not expected to pose a significant
bioaccumulation hazard. | [Incompatibilities]
Self-reactive. May form explosive peroxides on exposure to air. High heat can cause a violent chemical reaction that will cause container rupture. Fires,explosions, or hazardous polymerization may result fromcontact with air, strong oxidizers, strong acids, ozone, rust,nitrogen dioxide, phenol, chlorine dioxide, crotonaldehyde,or a free radical polymerization initiator, such as hydroquinone. Unsafe in contact with acetylide-forming materials,such as monel, copper, and copper alloys (piping material for this gas must not contain more than 63% copper). Addinhibitor (such as tributylcatechol) to prevent self-polymerization and monitor to insure effective levels are maintainedat all times. May accumulate static electrical charges, andmay cause ignition of its vapors. | [Toxics Screening Level]
The Initial Threshold Screening Level (ITSL) for 1,3-butadiene (BD) is 33 μg/m3 with annual averaging time (AT). | [Waste Disposal]
Disposal of l,3-butadiene by venting, incineration,
using a suitable flare system, or by other
means may be subject to permitting by federal,
state, provincial, or local regulations. Persons
involved with disposal of 1,3-butadiene should
check with the environmental authorities having
jurisdiction to determine the applicability of
permitting regulations to disposal activities. | [GRADES AVAILABLE]
1,3-Butadiene is available for commercial and
industrial use in various grades having much the
same component proportions from one producer
to another.
All grades contain approximately lIS ppm of
a polymerization inhibitor, such as tertiary-
butylcathechol. Distillation or washing with
dilute caustic solution is used for removing the
inhibitor when necessary. |
| Safety Data | Back Directory | [Hazard Codes ]
F+,T,F,N | [Risk Statements ]
R45:May cause cancer.
R12:Extremely Flammable.
R67:Vapors may cause drowsiness and dizziness.
R65:Harmful: May cause lung damage if swallowed.
R63:Possible risk of harm to the unborn child.
R48/20:Harmful: danger of serious damage to health by prolonged exposure through inhalation .
R36/38:Irritating to eyes and skin .
R11:Highly Flammable.
R62:Possible risk of impaired fertility.
R51/53:Toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment .
R38:Irritating to the skin. | [Safety Statements ]
S53:Avoid exposure-obtain special instruction before use .
S45:In case of accident or if you feel unwell, seek medical advice immediately (show label where possible) .
S62:If swallowed, do not induce vomiting: seek medical advice immediately and show this container or label .
S46:If swallowed, seek medical advice immediately and show this container or label .
S36/37:Wear suitable protective clothing and gloves .
S26:In case of contact with eyes, rinse immediately with plenty of water and seek medical advice .
S61:Avoid release to the environment. Refer to special instructions safety data sheet .
S33:Take precautionary measures against static discharges .
S16:Keep away from sources of ignition-No smoking . | [RIDADR ]
UN 1010 2.1
| [WGK Germany ]
2
| [RTECS ]
EI9275000
| [F ]
4.5-31 | [Autoignition Temperature]
788 °F | [Hazard Note ]
Extremely Flammable/Carcinogen | [DOT Classification]
2.1 (Flammable gas) | [HazardClass ]
2.1 | [PackingGroup ]
II | [HS Code ]
29012410 | [Safety Profile]
Confirmed carcinogen
with experimental carcinogenic and
neoplastigenic data. An experimental
teratogen. Mutation data reported.
Inhalation of high concentrations can cause
unconsciousness and death. Human
systemic effects by inhalation: cough,
hallucinations, dstorted perceptions,
changes in the visual field and other | [Hazardous Substances Data]
106-99-0(Hazardous Substances Data) | [Toxicity]
LC50 (inhalation) for mice 270 gm/m3/2-h, rats 285 gm/m3/4-h (quoted, RTECS, 1985). | [IDLA]
2,000 ppm (10% LEL) |
| Raw materials And Preparation Products | Back Directory | [Raw materials]
Toluene-->Nitrogen-->Acetonitrile-->PETROLEUM ETHER-->Calcium carbide-->n-Butane | [Preparation Products]
Sulfolane-->Hexamethylenediamine-->Styrene Butadiene Rubber-->DOWEX(R) 1X8-->4-Vinylcyclohexene dioxide-->FUSEL OIL-->1-BUTENE-->N-Hydroxymethyl-3,4,5,6-tetrahydrophthalimide-->2-(2-CHLOROETHOXY)-BENZENESULFONAMIDE-->Captan-->Adiponitrile-->Menadione-->cis-1,2,3,6-Tetrahydrophthalic anhydride-->Maleic hydrazide-->Cross-linking agent-->1,2,5,6,9,10-Hexabromocyclododecane-->2-BUTANOL-->Dodecanedioic acid-->5-ETHYLIDENE-2-NORBORNENE-->N,N,N',N'-TETRAMETHYL-2-BUTENE-1,4-DIAMINE-->Methacrylonitrile-->modified filling agent SBR-->CYCLODODECANE-->1,2,3,6-Tetrahydrophthalic anhydride-->Muscone-->POLYBUTADIENE DIACRYLATE-->ABS Resins-->1,4-HEXADIENE-->3-SULFOLENE-->Polyvinylpyrrolidone-->butadiene resin emulsion LHYJ-DS50-->Cyclododeca-1,5,9-triene-->2,4,4-TRIMETHYL-1-PENTENE-->Ti(Co)Ziegler catalyst-->Carboxy styrene-butadiene latex-->IRONE-->butadiene-styrene latex-->VITAMIN K4-->1,2,3,4-Tetrabromobutane |
|
|