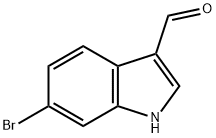
6-Bromoindole-3-carboxaldehyde synthesis
- Product Name:6-Bromoindole-3-carboxaldehyde
- CAS Number:17826-04-9
- Molecular formula:C9H6BrNO
- Molecular Weight:224.05

52415-29-9
415 suppliers
$9.00/1g
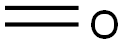
50-00-0
892 suppliers
$10.00/25g

17826-04-9
182 suppliers
$10.00/250mg
Yield:17826-04-9 73%
Reaction Conditions:
with iron(III) chloride;ammonia in water;N,N-dimethyl-formamide at 130; for 5 h;
Steps:
Indolecarbaldehydes 2a-2aa; General Procedure
General procedure: A 50 mL round-bottomed flask equipped with a magnetic stirringbar was charged with the appropriate indole 1 (0.5 mmol,1.0 equiv), 37% aq HCHO (0.5 mmol, 0.0406 g, 1.0 equiv), 25% aqNH3 (1.0 mmol, 0.0681 g, 2.0 equiv), FeCl3 (0.01 mmol, 0.0016 g,2 mol%), and DMF (2 mL). The flask was fitted with a reflux condenser,and the mixture was stirred at 130 °C under open air.When the reaction was complete (TLC), the mixture was cooledto r.t., diluted with sat. aq NaCl (10 mL) and 0.5 M aq HCl (2 mL),and extracted with EtOAc (3 x 7 mL). The organic layers werecombined, washed with sat. aq NaHCO3 (10 mL) and sat. aq NaCl(10 mL), dried (Na2SO4), and concentrated under reduced pressure.The residue was purified by flash column chromatography(silica gel, hexane-EtOAc).
References:
Wang, Qing-Dong;Zhou, Bin;Yang, Jin-Ming;Fang, Dong;Ren, Jiangmeng;Zeng, Bu-Bing [Synlett,2017,vol. 28,# 19,p. 2670 - 2674] Location in patent:supporting information

52415-29-9
415 suppliers
$9.00/1g
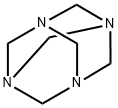
100-97-0
0 suppliers
$14.00/250G

17826-04-9
182 suppliers
$10.00/250mg

52415-29-9
415 suppliers
$9.00/1g
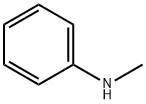
100-61-8
406 suppliers
$8.00/1g

17826-04-9
182 suppliers
$10.00/250mg
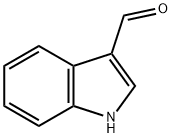
487-89-8
623 suppliers
$6.00/10g
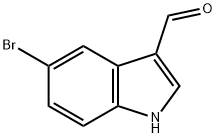
877-03-2
234 suppliers
$6.00/1g

17826-04-9
182 suppliers
$10.00/250mg
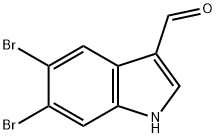
17900-95-7
24 suppliers
inquiry
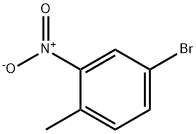
60956-26-5
297 suppliers
$8.00/10g

17826-04-9
182 suppliers
$10.00/250mg