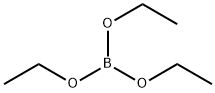
??????
|
|
?????? ??
- ???
- -84.5 °C
- ?? ?
- 117-118 °C(lit.)
- ??
- 0.858 g/mL at 25 °C(lit.)
- ?? ??
- 5.04 (vs air)
- ???
- n
20/D 1.374(lit.)
- ???
- 52 °F
- ?? ??
- 0-6°C
- ???
- ??? ? ???? ?? ??.
- ??? ??
- ??
- Specific Gravity
- 0.864
- ??
- ???
- ???
- ??? ????
- Hydrolytic Sensitivity
- 7: reacts slowly with moisture/water
- ??
- Moisture Sensitive
- BRN
- 1699052
- CAS ??????
- 150-46-9(CAS DataBase Reference)
??
- ?? ? ?? ??
- ?? ? ???? ?? (GHS)
| ??? ?? | F | ||
|---|---|---|---|
| ?? ???? ?? | 11 | ||
| ????? | 7-16-23-9-33 | ||
| ????(UN No.) | UN 1176 3/PG 2 | ||
| WGK ?? | 2 | ||
| RTECS ?? | ED5075000 | ||
| F ?????? | 21 | ||
| TSCA | Yes | ||
| ?? ?? | 3 | ||
| ???? | II | ||
| HS ?? | 29209085 | ||
| ?? | eye-rbt 100 mg MLD 14KTAK -,706,64 | ||
| ???? ?? | 97-3-40 |
?????? C??? ??, ??, ??
??? ??
Triethyl borate is a Colorless liquid; mild odor. Hydrolyzes rapidly depositing boric acid in finely divided crystalline form.??
Antiseptics, disinfectants, antiknock agent.Triethyl borate is used as a solvent in resins, paints, varnishes, adhesives and sealant chemicals. It is used as a catalyst of synthetic waxes and resins. It is an active component of flame retardants in the textile industry and in welding fluxes. It is used to prepare m-tolyl-acetic acid ethyl ester by reacting with 1-bromomethyl-3-methyl-benzene and carbon monoxide using as 1,5-hexadiene dium chloride as a catalyst. Further, it is used in pyrotechnics.
Triethyl borate may be used as boron source to synthesize lithium borophosphates, alumina borosilicate ceramic nanofibres and boron-doped single wall carbon nanotubes.CVD precursor for boron modified SiO2 in microelectronics Intermediate for organic-modified borosiloxanes.
??
ChEBI: A member of the class of borate esters obtained by the formal condensation of three equivalents of ethanol with boric acid.?? ??
Triethyl borate can be prepared by adding anhydrous ethanol with boric acid or boron trioxide. Can be purified via distillation.???
Flammable, dangerous fire risk.Safety Profile
Moderately toxic by ingestion. Aneye irritant. A very dangerous fire hazard when exposed toheat or flame. When heated to decomposition it emitsacrid smoke and irritating fumes.Purification Methods
Dry it over sodium, then distil it. Also fractionate it through a gauze packed column. [Charnlet et al. J Chem Soc 2288 1952, as for tributyl borate Johnson & Tompkins Org Synth Coll Vol II 106 1943, Beilstein 1 III 1339, 1 IV 1365.]?????? ?? ?? ? ???
???
?? ??
2-Furanboronic acid
4-Fluoronaphtalene-1-boronic acid
PHENYL-D5-BORONIC ACID
10-(2-Naphthyl) anthracene-9-boronic ?
2,6-DICHLOROPHENYLBORONIC ACID
???????
3-(9H-????-9-?)?????
6-(???????)-2-???
2-(??????)?????
??[C][1,2,5]?????-5-???
6,9- ?? ?? -9H- ?? ? -3- ? -3- ?? ?
???,[4-CHLORO-3-(TRIFLUOROMETHOXY)??]-
?????? ?? ??
???( 248)?? ??
| ??? | ?? | ??? | ?? | ?? ? | ?? |
|---|---|---|---|---|---|
| Hebei Chuanghai Biotechnology Co., Ltd | +8615531157085 |
abby@chuanghaibio.com | China | 8808 | 58 |
| Henan Fengda Chemical Co., Ltd | +86-371-86557731 +86-13613820652 |
info@fdachem.com | China | 20235 | 58 |
| Shanghai Daken Advanced Materials Co.,Ltd | +86-371-+86-371-66670886 |
info@dakenam.com | China | 19805 | 58 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 |
info@tianfuchem.com | China | 21628 | 55 |
| career henan chemical co | +86-0371-86658258 +8613203830695 |
sales@coreychem.com | China | 29858 | 58 |
| Hubei xin bonus chemical co. LTD | 86-13657291602 |
linda@hubeijusheng.com | CHINA | 22963 | 58 |
| Shandong chuangyingchemical Co., Ltd. | 18853181302 |
sale@chuangyingchem.com | CHINA | 5906 | 58 |
| CONIER CHEM AND PHARMA LIMITED | +8618523575427 |
sales@conier.com | China | 49732 | 58 |
| Antai Fine Chemical Technology Co.,Limited | 18503026267 |
info@antaichem.com | CHINA | 9636 | 58 |
| Hebei Mujin Biotechnology Co.,Ltd | +86 13288715578 +8613288715578 |
sales@hbmojin.com | China | 12809 | 58 |