Flumazenil Chemische Eigenschaften,Einsatz,Produktion Methoden
R-S?tze Betriebsanweisung:
R36/37/38:Reizt die Augen, die Atmungsorgane und die Haut.
S-S?tze Betriebsanweisung:
S26:Bei Berührung mit den Augen sofort gründlich mit Wasser abspülen und Arzt konsultieren.
S27:Beschmutzte, getr?nkte Kleidung sofort ausziehen.
S36/37/39:Bei der Arbeit geeignete Schutzkleidung,Schutzhandschuhe und Schutzbrille/Gesichtsschutz tragen.
Beschreibung
Flumazenil is a benzodiazepine antagonist useful as a fast-acting antidote in the treatment
of benzodiazepine intoxication, and in reversing the central sedative effects of
benzodiazepines during anesthesia.
Chemische Eigenschaften
Flumazenil is a white to off-white crystalline compound with an octanol:buffer partition coefficient of 14 to 1 at pH 7.4. It is insoluble in water but slightly soluble in acidic aqueous solutions.
Verwenden
Flumazenil is an imidazodiazepine which selectively blocks the central effects of classic benzodiazepines. It is used as benzodiazepine antagonist sedation reversal drug.
Definition
ChEBI: Flumazenil is an organic heterotricyclic compound that is 5,6-dihydro-4H-imidazo[1,5-a][1,4]benzodiazepine which is substituted at positions 3, 5, 6, and 8 by ethoxycarbonyl, methyl, oxo, and fluoro groups, respectively. It is used as an antidote to benzodiazepine overdose. It has a role as a GABA antagonist and an antidote to benzodiazepine poisoning. It is an ethyl ester, an organofluorine compound and an imidazobenzodiazepine.
synthetische
The Synthesis of Flumazenil
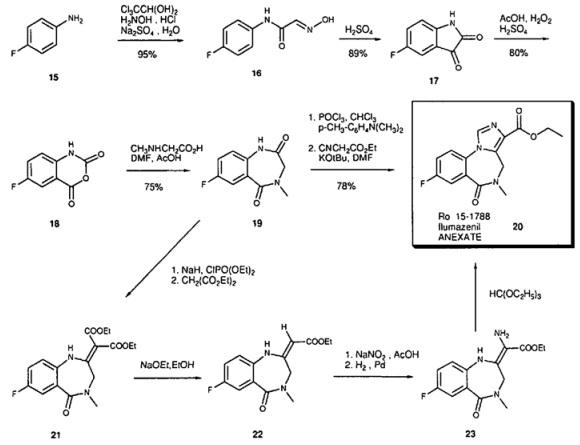
Starting with 4-fluoroaniline (15) the isatin 17 is synthesized via the Sandmeyer synthesis; isatin is then oxidized with peracetic acid to the isatoic anhydride 18. Reaction with sarcosine in DMF leads to the benzodiazepine-2,5-dione 19. This is converted to the iminochloride by reaction with POCI3 . In the key step the imidazoester is built up by reaction with deprotonated ethyl isocyanoacetate [8]. Since ethyl isocyanoacetate is not very stable, an alternative synthesis based on the synthesis of midazolam was developed for large scale-production. Tnthis synthesis diethylmalonate is used. The diester 21 is then transformed to the monoester 22 hy deethoxycarbonylation. Nitrosation and catalytic reduction lead to the amino compound 23. The final carbon atom is introduced by reaction with the orthoester.
Biologische Aktivit?t
Flumazenil is a GABAA receptor antagonist with non-selective for α 1, α 2, α 3 or α 5 (IC50 = 2 nM in a radioligand binding assay using rat cortical synaptosomes). Flumazenil also acts as a partial agonist of GABAA receptors, decreasing the amplitude of electrically stimulated population spikes in rat hippocampal CA1 pyramidal neurons. It increases the number of entries into the open arms of the elevated plus maze in high-anxiety BALB/c, but not C57BL/6, mice when administered at doses ranging from 0.1 to 1,000 μg/kg. Flumazenil (5 and 10 mg/kg) prevents a reduction in burying behavior induced by the GABAA receptor positive allosteric modulator allopregnanolone in ovariectomized rats when administered at doses of 5 and 10 mg/kg. Formulations containing flumazenil have been used to reverse sedation induced by benzodiazepines and in the treatment of benzodiazepine overdose or withdrawal.
Pharmakokinetik
Flumazenil is a competitive antagonist at the GA BAA benzodiazepine
binding site for all other ligands. I t rapidly reverses the CN S and dangerous
physiological effects of benzodiazepines following iatrogenic overdose or
deliberate self-harm. I t has no effect on benzodiazepine metabolism.
Flumazenil is rapidly cleared from plasma and metabolised by the liver and
has a very short elimination half-life (<1h). Its duration of action depends on
the dose administered and the duration of action of the drug to be
antagonised; repeated administration or infusions may be necessary.
Mode of action
Flumazenil, an imidazobenzodiazepine derivative, antagonizes the actions of benzodiazepines on the central nervous system. Flumazenil competitively inhibits the activity at the benzodiazepine recognition site on the GABA/benzodiazepine receptor complex. In animal experiments the effects of compounds showing no affinity for the benzodiazepine receptor, e.g. barbiturates, ethanol, meprobamate, GABA mimetics, adenosine receptor agonists and other agents were not affected by flumazenil, but those of nonbenzodiazepine agonists of benzodiazepine receptors, such as cyclopyrrolones (e.g. zopiclone) and triazolopyridazines were blocked.
Einzelnachweise
Flumazenil in benzodiazepine overdose
DOI:
10.1503/cmaj.160357Pharmacological uses of flumazenil in benzodiazepine use disorders: a systematic review of limited data
DOI:
10.1177/0269881120981390
Flumazenil Upstream-Materialien And Downstream Produkte
Upstream-Materialien
Downstream Produkte

