exo-1,7,7-Trimethylbicyclo[2.2.1]hept-2-ylmethacrylat Chemische Eigenschaften,Einsatz,Produktion Methoden
R-S?tze Betriebsanweisung:
R36/37/38:Reizt die Augen, die Atmungsorgane und die Haut.
S-S?tze Betriebsanweisung:
S26:Bei Berührung mit den Augen sofort gründlich mit Wasser abspülen und Arzt konsultieren.
S36:DE: Bei der Arbeit geeignete Schutzkleidung tragen.
Beschreibung
Isobornyl methacrylate (IBOMA) is an acrylate monomer with a bridging ring structure. It can be used in polymerisation reactions to make other coatings or plastic products. Advantages of IBOMA and its polymers include: high viscosity, good compatibility, low volatility, low toxicity, UV resistance, water resistance, chemical resistance, and low adverse biological effects.
Chemische Eigenschaften
CLEAR COLORLESS TO YELLOW LIQUID
Charakteristisch
Isobornyl methacrylate is a colorless transparent liquid. It is a monomer that unifies hardness and flexibility. Due to its molecular structure, its polymer has excellent high gloss, sharp image, scratch resistance, medium resistance and weather resistance, and Its hygroscopicity is significantly lower than that of MMA (methyl methacrylate). In addition, IBOMA-added acrylic resin has good compatibility with polyester, alkyd and many volatile paint film-forming substances.
Verwenden
Isobornyl methacrylate is a versatile hydrophobic monomer with a good combination of hardness and flexibility that can improve the chemical and water resistance of polymer systems.
synthetische
Isobornyl methacrylate was synthesized by direct esterification of camphene and methacrylic acid.
Allgemeine Beschreibung
Isobornyl methacrylate is a reactive solvent with low vapor pressure. It facilitates the free radical polymerization by forming a polymer with high glass transition temperature (T
g).
Synthese
Isobornyl methacrylate is obtained by reacting (1S,2S,4S)-(+)-isoborneol and 4-methoxyphenol.Dissolve5g (1S,2S,4S)-(+)-isoborneol in 15 mL toluene. Add 4 mg (0.032 mmol) 4-methoxyphenol to the solution. After the addition of 0.4 g (3.24 mmol)4-dimethylamino pyridine, add a solution of 10 g (64.8 mmol) methacrylic anhydride (MAA) in 10 mL toluene dropwise to the mixture. Heat the reaction mixture to 80 °C. Stir the reaction mixture for 16 hours. After cooling down to room temperature, wash the mixture with 50 mL 1M sodium bicarbonate solution four times. Wash the organic phase with 50 mL brine two times. Separate the organic phase. Remove the toluene by rotary evaporation followed by a fractional distillation under reduced pressure.
1H NMR (CDCl3, 300 MHz): δ(ppm) = 6.06 (dq, 1H, 1); 5.51 (dq, 1H, 2); 4.71 (m ,1H, 4); 1.93 (dd, 3H, 3); 1.87-1.5 (m, 5H, 6,7,8); 1.23-1.04 (m, 2H, 5); 1.012 (s, 3H, 9);0.86 (s, 3H, 10); 0.85 (s, 3H, 11). IR (film): ν(cm-1) = 2954 (CH2), 2879 (CH2), 1715 (C=O) 1454 (CH2), 1157 and 1053 (C=O).
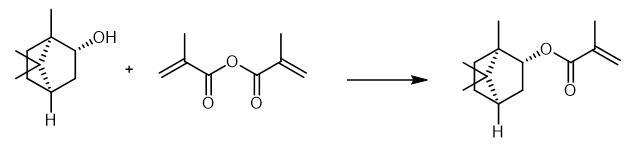
Fig The synthetic method of Isobornyl methacrylate
exo-1,7,7-Trimethylbicyclo[2.2.1]hept-2-ylmethacrylat Upstream-Materialien And Downstream Produkte
Upstream-Materialien
Downstream Produkte

