Biphenyl-4-ol Chemische Eigenschaften,Einsatz,Produktion Methoden
R-S?tze Betriebsanweisung:
R36/37/38:Reizt die Augen, die Atmungsorgane und die Haut.
R51/53:Giftig für Wasserorganismen, kann in Gew?ssern l?ngerfristig sch?dliche Wirkungen haben.
R38:Reizt die Haut.
S-S?tze Betriebsanweisung:
S26:Bei Berührung mit den Augen sofort gründlich mit Wasser abspülen und Arzt konsultieren.
S36/37:Bei der Arbeit geeignete Schutzhandschuhe und Schutzkleidung tragen.
S61:Freisetzung in die Umwelt vermeiden. Besondere Anweisungen einholen/Sicherheitsdatenblatt zu Rate ziehen.
S36:DE: Bei der Arbeit geeignete Schutzkleidung tragen.
Chemische Eigenschaften
light tan solid (odour threshold detection limit 0.7 ppm)
Verwenden
Fluorescence and phosphorescence quantum yields and fluorescence and phosphorescence lifetimes were obtained for 4-phenylphenol adsorbed on filter paper with either NaCl, NaBr, or NaI at 296 and 93 K. The solid-surface fluorescence and phosphorescence quantum yield values and phosphorescence lifetime values were obtained for p-aminobenzoic acid (PABA) and 4-phenylphenol adsorbed on α-cyclodextrin/NaCl mixtures. 4-Phenylphenol is used as an antioxidant and is a potential EDC.
synthetische
4-phenylphenol synthesis: Add to a 50 ml round-bottom flask, in this order, 122 mg of phenylboronic acid, 414 mg of potassium carbonate, 220 mg of 4-iodophenol, and 10 ml of deionized water. Weigh in a suitably sized container 3 mg Pd on C 10%, add 1 ml of deionized water, and stir gently by hand to form a slurry that is then transferred to the reaction flask.
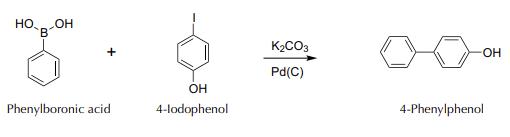
Couple the flask to a water-jacketed condenser, and reflux the mixture on a hot plate with a magnetic stirrer vigorously for 30 min (until a precipitate appears). After this time, switch off the plate and allow to cool to r.t. Add HCl 2 M to an acidic pH (check with indicator paper). Separate the resulting solid, still containing the catalyst, by filtering with a Hirsch funnel. Wash the solid with 10 ml of water. Then, in a Hirsch funnel, add 10 ml of MeOH, and collect the filtrate in a clean container. Add to the resulting MeOH solution 10 ml of deionized water to obtain the precipitate of the product. Purify by recrystallization, heating in a water bath container with the precipitate and the MeOH/H2O mixture. If necessary, add 1 to 2 ml more of hot MeOH, to finish dissolving the solid. Filter under vacuum with a Hirsch funnel, air dry the solid (can recover the next day). Weigh and calculate the yield.
Definition
ChEBI: 4-Phenylphenol is a member of the class of hydroxybiphenyls that is biphenyl carrying a hydroxy group at position 4.
Allgemeine Beschreibung
4-Phenylphenol undergoes enzymatic polymerization and polymer developed is characterized by matrix-assisted laser desorption ionization time-of-flight mass spectrometry. It is the intermediate in manufacture of 4-alkyl substituted phenol-formaldehyde resins.
Sicherheitsprofil
Acute poison by intraperitonealroute. Questionable carcinogen with experimentalcarcinogenic and tumorigenic data. When heated todecomposition it emits acrid, irritating fumes.
l?uterung methode
Crystallise the phenol from aqueous EtOH, *C6H6, and dry it in a vacuum over CaCl2 [Buchanan et al. J Am Chem Soc 108 7703 1986]. [Beilstein 6 IV 4600.]
Biphenyl-4-ol Upstream-Materialien And Downstream Produkte
Upstream-Materialien
Downstream Produkte

