| Identification | More | [Name]
Mezlocillin | [CAS]
51481-65-3 | [Synonyms]
3,3-dimethyl-7-[[2-[(3-methylsulfonyl-2-oxo-imidazolidin-1-yl)carbonylamino]-2-phenyl-acetyl]amino]-6-oxo-2-thia-5-azabicyclo[3.2.0]heptane-4-carboxylic acid
MEZLOCILLIN
(2s-(2-alpha,5-alpha,6-beta)(s*))-sodiumsal
4-thia-1-azabicyclo(3.2.0)heptane-2-carboxylicacid,3,3-dimethyl-6-(((((3-(met
antibioticbay-f1353
Melocillin sodium
Meloxacam acid
Mezlocillin acid
MEZLOCILLIN SODIUM USP(CRM STANDARD)
3,3-Dimethyl-7-[[2-[(3-methylsulfonyl-2-oxo-imidazolidin-1-yl)carbonylamino]-2-phenyl-acetyl]amino]-6-oxo-2-thia-5-azabicyclo[3.2.0]heptane-4-carboxylic acid
6α-[[(R)-[[[3-(Methylsulfonyl)-2-oxoimidazolidin-1-yl]carbonyl]amino]phenylacetyl]amino]penicillanic acid
Multocillin
Mezlocillin(Mezlocillin sodium) | [EINECS(EC#)]
257-233-8 | [Molecular Formula]
C21H25N5O8S2 | [MDL Number]
MFCD00056866 | [Molecular Weight]
539.58 | [MOL File]
51481-65-3.mol |
| Hazard Information | Back Directory | [Description]
Mezlocilin is an acylaminopenicillin, which has been
shown to cause both immediate and delayed hypersensitivity
in a nurse. | [Originator]
Baypen,Bayer,W. Germany,1977 | [Uses]
Antibacterial. | [Uses]
Like azlocillin, mezlocillin is used for infections of the urinary tract, gynecological infec�tions, intraabdominal infections, skin infections, and respiratory tract infections. Synonyms
of this drug are baypen, mezlin, and optocillin. | [Uses]
Mezlocillin is a broad spectrum penicillin antibiotic. | [Definition]
ChEBI: A penicillin in which the substituent at position 6 of the penam ring is a (2R)-2-[3-(methanesulfonyl)-2-oxoimidazolidine-1-carboxamido]-2-phenylacetamido group. | [Manufacturing Process]
9.3 parts by weight of ampicillin were suspended in 80% strength aqueous
tetrahydrofuran (140 parts by volume) and sufficient triethylamine
(approximately 6.3 parts by volume) was added dropwise while stirring at
20°C, just to produce a clear solution and to give a pH value of between 7.5
and 8.2 (glass electrode). The mixture was cooled to 0°C and 5.1 parts by
weight of 3-methylsulfonyl-imidazolidin-2-one-1-carbonyl chloride were added
gradually in portions over the course of 30 minutes, while the mixture was
stirred and kept at a pH value of between 7 and 8 by simultaneous addition of
triethylamine.
The carbonyl chloride reactant was prepared by reacting 2-imidazolidone with
methanesulfonyl chloride then that product with phosgene. The mixture was
stirred for 10 minutes at 0°C and subsequently further stirred at room
temperature until no further addition of triethylamine was necessary to
maintain a pH value of 7 to 8. 150 parts by volume of water were added andthe tetrahydrofuran was largely removed in a rotary evaporator at room
temperature.
The residual aqueous solution was extracted once by shaking with ethyl
acetate, covered with 250 parts by volume of fresh ethyl acetate and acidified
to pH 1.5 to 2.0 with dilute hydrochloric acid while being cooled with ice. The
organic phase was separated off, washed twice with 50 parts by volume of
water at a time and dried for 1 hour over anhydrous MgSO4 in a refrigerator.
After filtration, about 45 parts by volume of a 1 molar solution of sodium 2-
ethylhexanoate in ether containing methanol were added to the solution of the
penicillin. The mixture was concentrated on a rotary evaporator until it had an
oily consistency and was dissolved in a sufficient amount of methanol by
vigorous shaking, and the solution was rapidly added dropwise, with vigorous
stirring, to 500 parts by volume of ether which contained 10% of methanol.
The precipitate was allowed to settle for 30 minutes, the solution was
decanted from the precipitate, and the latter was again suspended in ether,
filtered off and washed with anhydrous ether. After drying over P2O5 in a
vacuum desiccator, the sodium salt of the mezlocillin was obtained in the form
of a white solid substance. | [Brand name]
Multocillin (Bayer). | [Therapeutic Function]
Antibiotic | [Antimicrobial activity]
A semisynthetic acylureidopenicillin supplied as the sodium
salt for parenteral administration.
Ampicillin-susceptible strains of H. influenzae and Neisseria
spp. are very susceptible, but β-lactamase-producing organisms
are usually resistant. It is less active than azlocillin and
piperacillin against Ps. aeruginosa and has variable activity
against B. fragilis, independent of β-lactamase production. It
exhibits typical β-lactam synergy with aminoglycosides against
Ps. aeruginosa and enterobacteria.
It attains peak concentrations of 250 mg/L after a 2 g intravenous
infusion, with a plasma half-life of 55 min. Protein
binding is 20–30%. It distributes into multiple tissues and
human body fluids at therapeutically useful concentrations.
Up to 60% of the dose is recoverable unchanged from the
urine, with up to 2.5% excreted in the bile.
Toxicity and side effects are similar to those associated
with carboxypenicillins. Its clinical use is for serious infections
with susceptible organisms, including lower respiratory tract,
intra-abdominal, urinary tract and gynecological infections.
Commercial availability is quite limited. | [General Description]
Mezlocillin was synthesized by Bayer in 1974. The NH2 residue of ampicillin was acylated. Mezlocillin is a member of the so-called ureidopenicillins and shows two- to eightfold greater activity against Citrobacter, Enterobacter, Klebsiella, Escherichia coli, and Haemophilus influenzae than ampicillin, carbenicillin, or sulbenicillin. Its activity against gram-positive bacteria is almost the same as that of carbenicillin. Mezlocillin is used by intravenous administration for therapy of sepsis, meningitis, and respiratory tract, urinary tract, and abdominal infections. | [Synthesis]
Mezlocillin, (2S,5R,6R)-3,3-dimethyl-7-oxo-6-[(R)-2-[(3-methylsulfonyl)-2-
oxoimidazolidin-1-carboxamido]-2-phenylacetamido]-4-thia-1-azabicyclo[3.2.0]-heptan-2-
carboxylic acid (32.1.1.27), is synthesized by acylating ampicillin (32.1.1.16) with
3-chlorocarbonyl-1-methansulfonyl-2-imidazolidinone (32.1.1.26) in the presence of triethy�lamine. The necessary 3-chlorocarbonyl-1-methansulfonyl-2-imidazolidinone (32.1.1.26) is
synthesized by sulfonating 2-imidazolidinone with methanesulfonyl chloride, which forms 1-methanesulfonyl-2-imidazolidinone (32.1.1.25) and its subsequent reaction with phosgene.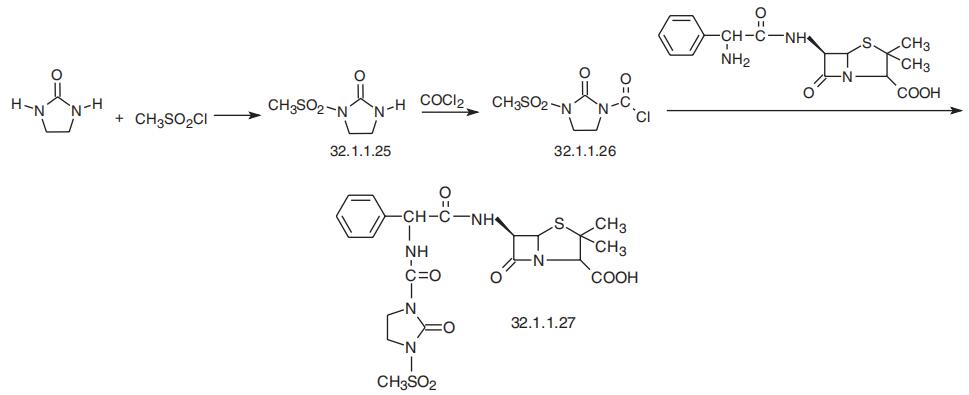
|
|
|