| Identification | More | [Name]
Titanocene dichloride | [CAS]
1271-19-8 | [Synonyms]
BIS(CYCLOPENTADIENYL)TITANIUM DICHLORIDE
BIS(CYCLOPENTADIENYL)TITANIUM(IV) DICHLORIDE
DICHLORODICYCLOPENTADIENYLTITANIUM
(DICYCLOPENTADIENE)TITANIUM CHLORIDE
DICYCLOPENTADIENYLTITANIUM DICHLORIDE
DI(CYCLOPENTADIENYL)TITANIUM(IV) DICHLORIDE
DYAD(DICYCLOPENTADIENE)TITANIUM CHLORIDE
TITANOCENE DICHLORIDE
bis(cyclopentadienyl)dichlorotitanium
bis(eta(5)-cyclopentadienyl)titaniumdichloride
Bis(eta(sup5)-cyclopentadienyl)titanium dichloride
bis(eta(sup5)-cyclopentadienyl)titaniumdichloride
bis(eta5-Cyclopentadienyl) titanium dichloride
Bis(pi-cyclopentadienyl)dichlorotitanium
Bis(pi-cyclopentadienyl)titanium dichloride
bis(pi-cyclopentadienyl)titaniumdichloride
bis(η-cyclopentadienyl)titaniumchloride
bis(η-cyclopentadienyl)-titaniumchloride
dichlorobis(1,3-cyclopentadiene)titanium
Dichlorobis(cyclopentadienyl)titanium | [EINECS(EC#)]
215-035-9 | [Molecular Formula]
C10H10Cl2Ti | [MDL Number]
MFCD00003723 | [Molecular Weight]
248.96 | [MOL File]
1271-19-8.mol |
| Chemical Properties | Back Directory | [Appearance]
Red crystals. Moderately
soluble in toluene, chloroform; sparingly soluble
in ether and water. Stable in dry air, slowly
hydrolyzed in moist air. | [Melting point ]
260-280 °C (dec.)(lit.)
| [Boiling point ]
355.52°C (estimate) | [density ]
1.6 g/mL at 25 °C(lit.)
| [vapor pressure ]
0.002Pa at 25℃ | [storage temp. ]
Inert atmosphere,Room Temperature | [solubility ]
slightly soluble in H2O, benzene; soluble in chloroform,ethanol, toluene | [form ]
crystal | [color ]
red | [Specific Gravity]
1.6 | [Stability:]
Stable. Incompatible with strong oxidizing agents. Decomposes in water. Moisture sensitive. | [Water Solubility ]
slow decomposition | [Hydrolytic Sensitivity]
4: no reaction with water under neutral conditions | [Sensitive ]
Air & Moisture Sensitive | [Merck ]
14,9482 | [LogP]
-1.35 at 20℃ | [CAS DataBase Reference]
1271-19-8(CAS DataBase Reference) | [NIST Chemistry Reference]
Titanocene dichloride(1271-19-8) | [EPA Substance Registry System]
1271-19-8(EPA Substance) |
| Safety Data | Back Directory | [Hazard Codes ]
Xi,T | [Risk Statements ]
R37/38:Irritating to respiratory system and skin .
R61:May cause harm to the unborn child.
R40:Limited evidence of a carcinogenic effect.
R34:Causes burns.
R33:Danger of cumulative effects.
R36/37/38:Irritating to eyes, respiratory system and skin . | [Safety Statements ]
S36:Wear suitable protective clothing .
S7/8:Keep container tightly closed and dry .
S45:In case of accident or if you feel unwell, seek medical advice immediately (show label where possible) .
S36/37/39:Wear suitable protective clothing, gloves and eye/face protection .
S26:In case of contact with eyes, rinse immediately with plenty of water and seek medical advice . | [RIDADR ]
UN3261 | [WGK Germany ]
3
| [RTECS ]
XR2050000
| [F ]
8-10-21 | [TSCA ]
Yes | [HazardClass ]
8 | [PackingGroup ]
II | [HS Code ]
29310095 | [Safety Profile]
Poison by intravenous
and intraperitoneal routes. Questionable
carcinogen with experimental neoplastigenic,
tumorigenic, and teratogenic data. Mutation
data reported. See also TITANIUM
COMPOUNDS. When heated to
decomposition it emits toxic fumes of Cl- | [Hazardous Substances Data]
1271-19-8(Hazardous Substances Data) | [Toxicity]
Titanocene dichloride may cause skin and mucous membrane irritation. The LD50 in mice has been reported as 60 mg/kg (i.p.) and 180 mg/kg (i.v.). In the rat, the LD50 is 25 mg/kg (i.p.). |
| Hazard Information | Back Directory | [General Description]
Red to red-orange crystals. | [Reactivity Profile]
TITANOCENE DICHLORIDE(1271-19-8) is incompatible with strong oxidizers. TITANOCENE DICHLORIDE(1271-19-8) may decompose on exposure to water. . | [Air & Water Reactions]
Stable in dry air. Decomposes in moist air and in water to form HCl . | [Fire Hazard]
Flash point data for this chemical are not available; however, TITANOCENE DICHLORIDE is probably combustible. | [Description]
Titanocene dichloride is an organotitanium compound with the chemical formula (η5-C5H5)2TiCl2, often written as Cp2TiCl2, which is a common reagent in organometallic chemistry and organic synthesis. Cp2TiCl2 does not form a "sandwich" structure like ferrocene, but a tetrahedral structure due to its four ligands around a metal center. Because of its anti-tumor activity, it has been used in clinical trials as a chemotherapeutic agent. | [Chemical Properties]
Red crystals. Moderately
soluble in toluene, chloroform; sparingly soluble
in ether and water. Stable in dry air, slowly
hydrolyzed in moist air. | [Chemical Properties]
Titanocene dichloride is irritating to the skin and mucous
membranes. | [Uses]
Catalyst; with aluminum alkyls as Ziegler-Natta polymerization catalyst. | [Uses]
Titanocene dichloride is used as an experimental cancer
chemotherapeutic agent. | [Preparation]
Cp2TiCl2 continues to be prepared similarly to its original synthesis by Wilkinson and Birmingham:
2 NaC5H5 + TiCl4 → (C5H5)2TiCl2 + 2 NaCl
The reaction is conducted in THF. Work-up entails extraction into chloroform/hydrogen chloride and recrystallization from toluene. In the original literature, the structure was poorly understood. Each of the two Cp rings are attached to Ti(IV) through all five carbon atoms. In organometallic chemical jargon, this bonding is referred to as η5 (see hapticity). | [Production Methods]
Titanocene dichloride is produced by the reaction of titanium
tetrachloride with cyclopentadienyl sodium. | [Reactions]
-
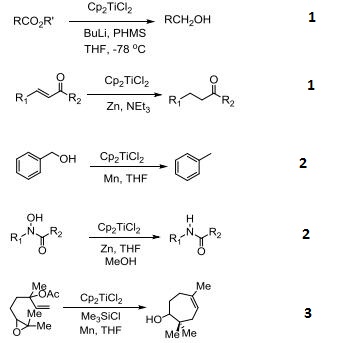
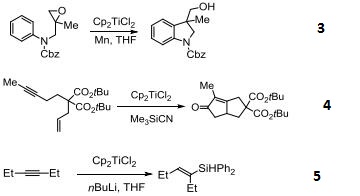
Precatalyst for reduction of esters and α,β-unsaturated ketones.
-
Catalyzes reductive deoxygenation of alcohols and hydroxylamines.
-
Catalyst for the radical cyclization of epoxides.
-
Reagent for the conversion of enynes to bicyclic cyclopentenones.
-
Catalyzes silylation of alkenes and alkynes.
| [Hazard]
Toxic by inhalation, irritant to skin and
mucous membranes. | [Flammability and Explosibility]
Nonflammable | [reaction suitability]
core: titanium
reagent type: catalyst | [Carcinogenicity]
Based on the results of 2 year
gavage studies, the National Toxicology Program determined
that there was equivocal evidence of the carcinogenic activity
of titanocene dichloride in male and female rats based on a
marginal increase in the incidence of forestomach squamous
cell effects. | [Carcinogenicity]
Titanocene administered by
intramuscular injection produced tumors both at the injection
site and at sites distant from the injection site.
Titanocene dichloride showed equivocal evidence of carcinogenicity
in male and female Fischer 244 rats in a 2-year
oral gavage study. | [Purification Methods]
It forms bright red crystals from toluene or xylene/CHCl3 (1:1) and sublimes at 190o/2mm. It is moderately soluble in EtOH and insoluble in Et2O, *C6H6, CS2, CCl4, pet ether and H2O. The crystalline dipicrate explodes on melting at 139-140o. [Wilkinson et al. J Am Chem Soc 75 1011 1953, IR: Wilkinson & Birmingham J Am Chem Soc 76 4281 1954, NMR and X-ray: Glivicky & McCowan Can J Chem 51 2609 1973, Clearfield et al. J Am Chem Soc 53 1622 1975, Beilstein 16 IV 1769.] | [Clinical claims and research]
Titanocene dichloride, [Ti(Z5 -C5H5)2Cl2], was the first organometallic transition metal compound to be investigated clinically as an anticancer agent. It contains a cis-dichloride motif as cisplatin and was selected from several early transition metal cyclopentadienyl complexes as the best candidate for further development. The chemistry of the hard Ti(IV) ion is different from that of Pt(II): for example, cisplatin binds preferentially to the N7 of guanine in DNA, whereas titanocene dichloride exhibits higher affinity for the phosphate backbone. [Ti(Z5 -C5H5)2Cl2] hydrolyzes quickly in water, yielding a solvated Ti(IV) ion with high affinity for transferrin. As for Ga(III), selective transport of titanium ions via the transferrin route appears plausible. In vitro, titanocene dichloride is active in cisplatin-resistant cancer cells. It entered clinical trials in 1993, revealing nephrotoxicity as dose-limiting side effect. In Phase II studies as singleagent therapy no advantage over other treatment regimens was observed, and the trials were thus abandoned. |
|
|