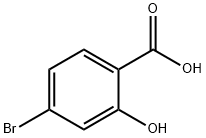
4-Bromo-2-hydroxybenzoic acid synthesis
- Product Name:4-Bromo-2-hydroxybenzoic acid
- CAS Number:1666-28-0
- Molecular formula:C7H5BrO3
- Molecular Weight:217.02
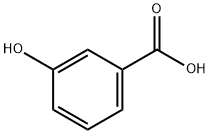
99-06-9
517 suppliers
$5.00/25g

1666-28-0
220 suppliers
$9.00/1g
Yield:1666-28-0 100%
Reaction Conditions:
with sulfuric acid;bromine in acetic acid
Steps:
I.92 [5-(4-Bromo-3-methoxy-phenyl)-3-methyl-2,3-dihydro-[1,3,4]thiadiazol-2-yl]-cyclohexyl-amine
[5-(4-Bromo-3-methoxy-phenyl)-3-methyl-2,3-dihydro-[1,3,4]thiadiazol-2-yl]-cyclohexyl-amine To a mixture of 3-hydroxybenzoic acid (14.480 mmol, 2 g) in acetic acid (14.5 mL) and sulfuric acid (1.5 mL) at 50° C., a solution of bromine (15.204 mmol, 0.780 mL) in acetic acid (7.2 mL) was added and stirred 30 minutes at 100° C. The reaction was allowed to cool to RT and diluted with water. The aqueous layer was extracted with ethyl acetate, washed with water and brine, dried (MgSO4), filtered, and concentrated under reduced pressure to give the 4-bromo-2-hydroxy-benzoic acid. Yield: 100%.
References:
Vergne, Fabrice;Ducrot, Pierre;Andrianjara, Charles;Bernardelli, Patrick;Lorthois, Edwige US2003/45557, 2003, A1

65-49-6
506 suppliers
$6.00/25g

1666-28-0
220 suppliers
$9.00/1g
22532-62-3
322 suppliers
$5.00/250mg

1666-28-0
220 suppliers
$9.00/1g
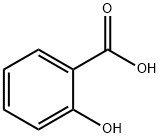
69-72-7
1290 suppliers
$5.00/10g

1666-28-0
220 suppliers
$9.00/1g