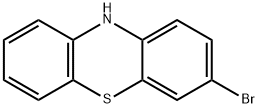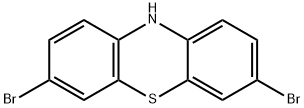
3,7-dibromo-10H-phenothiazine synthesis
- Product Name:3,7-dibromo-10H-phenothiazine
- CAS Number:21667-32-3
- Molecular formula:C12H7Br2NS
- Molecular Weight:357.06
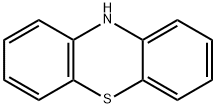
92-84-2
541 suppliers
$10.00/5g

21667-32-3
24 suppliers
inquiry
Yield: 100%
Reaction Conditions:
with bromine;acetic acid at 20; for 16 h;Inert atmosphere;
Steps:
1 Step 1: 3,7-Dibromo-1OH-phenothiazine
To a stirred solution of 10H-phenothiazine (5 g, 25.1 mmol)in AcOH (50 mL) was added Br2(3.3 mL, 63 mmol) and stirring continued at room temperature for 16 h. The reaction mixturewas filtered and dried to get the title compound asbrown solid(7 g, quantitative):’HNIVIR(DMSO 400 MHz) ? 6.55-6.57 (m, 2H),7.06-7.20 (m, 4H), 8.79 (s, 1H);MS (ESI) m/z358(M+2H).
References:
BIOIMICS AB;KIRSEBOM, Leif;UPADHAYAYA, Ram Shankar;KETHIRI, Raghava Reddy;VIRTANEN, Anders WO2017/222466, 2017, A1 Location in patent:Page/Page column 8; 9; 71; 72
![Ethanamine, N-[(3,5-dichlorophenyl)methylene]-2,2-diethoxy-](/CAS/20210305/GIF/1000210-73-0.gif)
1000210-73-0
0 suppliers
inquiry

21667-32-3
24 suppliers
inquiry
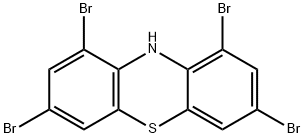
18621-99-3
0 suppliers
inquiry

21667-32-3
24 suppliers
inquiry