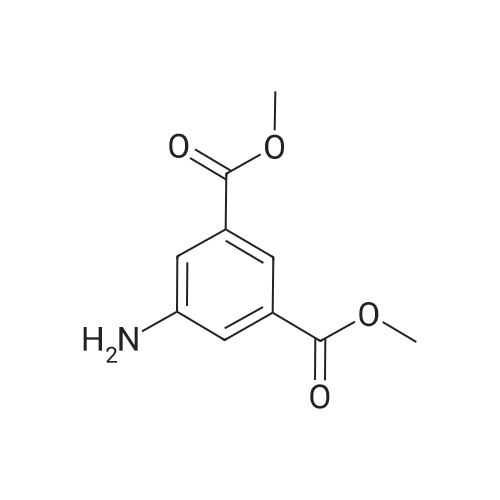
| 100% |
With ammonium formate;palladium 10% on activated carbon; In ethanol; at 40℃; for 2h; |
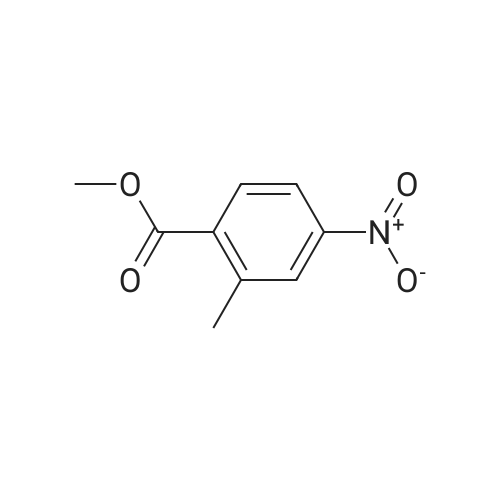
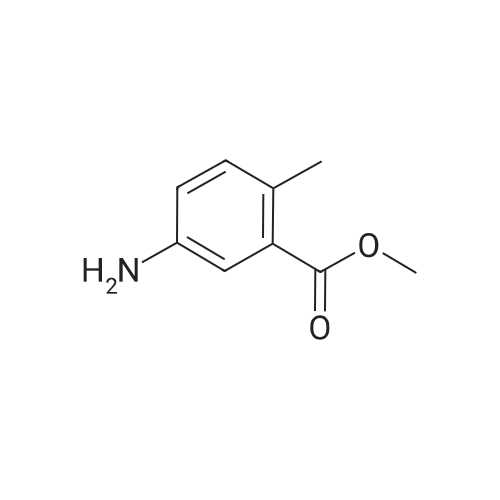
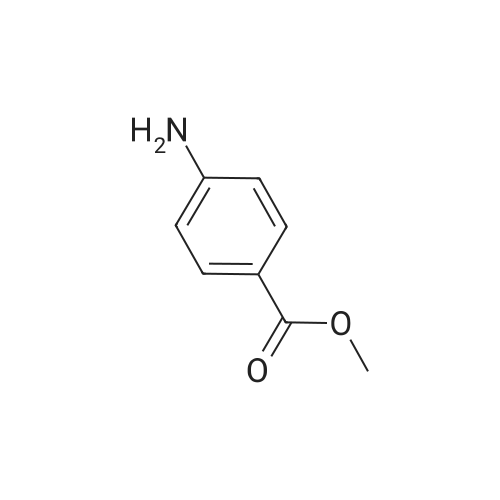
Intermediate 7; Methyl 4-amino-2-methylbenzoate; To a solution of <strong>[62621-09-4]methyl 2-methyl-4-nitrobenzoate</strong> (Intermediate 5) (3.25 g, 16.6 mmol) in ethanol was added Pd/C 10% (catalytic quantity) and ammonium formate (10.5 g, 0.17 mmol). The mixture was stirred at 400C for 2 hours. The mixture was filtered on celite, washed with diethyl ether. The filtrate was evaporated and the residue was diluted with diethyl ether, washed with water. The organic phase was dried over Na2SC>4, filtered and evaporated to give the title compound as brown oil (2.8 g, quantitative yield). NMR1H NMR (300 MHz), CDCI3 delta: 7.74 (d, 1 H, J=9.20 Hz), 6.41 (m, 2H), 3.75 (s, 3H), 2.47 (s, 3H). |
|
With iron(III) chloride; water; hydrazine; In ethanol; for 3h;Heating / reflux; Charcoal; |
Example 1: Synthesis of PDZ domain inhibitor (2-(l-HydroxypentvD-3-(2-phenvlethvl)-6-methvl)indole-5-carboxylic acid [Formula (I); Table 1A, compound 11; [0067] The general scheme for this synthesis was (Scheme D):; [0068] Methyl (4-amino-5-iodo-2-methyl)benzoate (4); A mixture of 2-iodo-5-nitrotoluene (1,10 g), triethylamine (16 mL), palladium acetate (68 mg), methanol (20 mL) and DMF (10 mL) was stirred at 90C overnight under carbon monoxide atmosphere (1 atm). The reaction mixture was diluted with ethyl acetate (200 mL), washed with water twice (100 mL each) followed by brine (100 mL), dried (Na2S04), and evaporated. The residue was filtered through a short pad of silica gel and evaporated to give 2 as a crude oil. A mixture of 2, ethanol (140 mL), water (2 mL), hydrazine monohydrate (3.8 mL), ferric trichloride (0.17 g) and charcoal (0.1 g) was stirred under reflux for 3 hours. The reaction mixture was filtrated, diluted with ethyl acetate (200 mL), washed with water twice (100 mL each) followed by brine (100 mL), dried (Na2S04), and evaporated. The residue was filtered through a short pad of silica gel and evaporated to give 3 as a crude oil. To a mixture of 3, methanol (33 mL), calcium carbonate (10 g), iodine monochloride solution (33 mL, 1M in dichloromethane) was added slowly at 4C and stirred overnight at the ambient temperature. The reaction mixture was filtrated, diluted with ethyl acetate (200 mL), washed with aqueous sodium sulfite solution (100 mL), followed by brine (100 mL), dried (Na2SC>4), and evaporated. The residue was purified by column chromatography (silica gel 0.2 L, eluent: 10% ethyl acetate in hexanes) and evaporated to give 4 (4.30 g) as pale brown crystals. *H NMR (CDC13, 400 MHz) 8 8.28 (s, 1H), 6.54 (s, 1H), 4,39 (broad s, 2H), 3.84 (s, 3H), 2.50 (s, 3H). |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 120K+ Compounds
120K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping