|
With morpholine; sulfur; In ethanol; at 50℃; for 18h; |
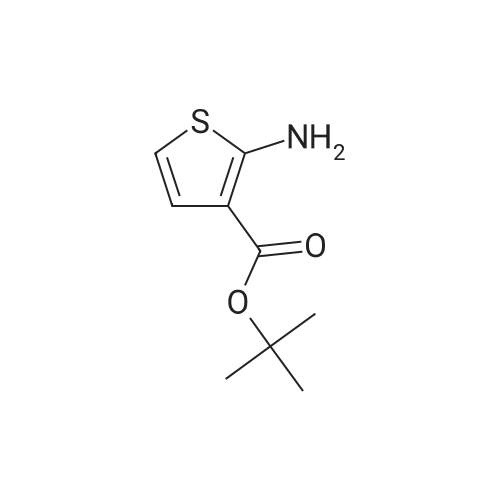
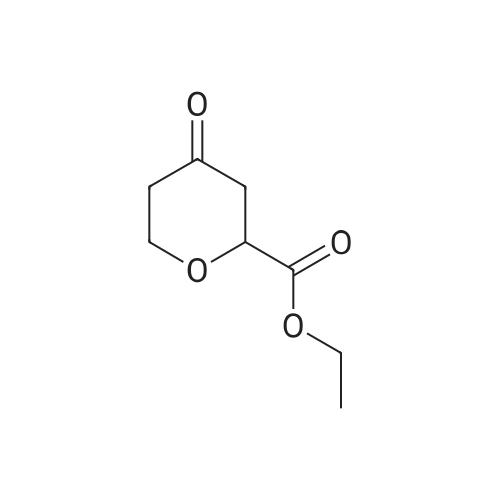
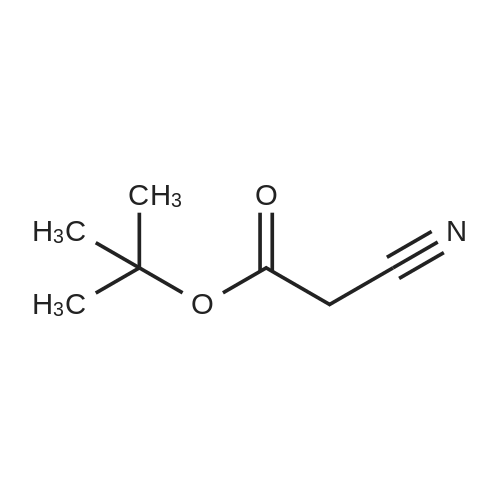
To a solution of 4-oxo-tetrahydro-2H-pyran-2-carboxylic acid ethyl (0.6 g, 3.5 mmol) in absolute ethanol (6 ml) was added sulfur (0.12 g, 3.85 mmol) and tert-butyl cyanoacetate (0.64 g, 4.55 mmol). The solution was stirred under nitrogen in a 50 C oil bath and morpholin (0.61 ml, 7.0 mmol) was added. The reaction was stirred for 18 hours and then cooled to ambient temperature and excess sulfur removed by filtration. The filtrate was concentrated in vacuo and reconstituted in ethyl acetate (50 ml). The organic phase was washed with brine (2 x 10 ml), dried (Na2SO4), filtered, and the solvent evaporated in vacuo . The residue was purified by silica gel chromatography using a gradient of ethyl acetate/hexane (20 to 25 % gradient) as eluent. Pure fraction of the two isomers were collected and the solvent evaporatedin vacuo which afforded 0.47 g of 2-amino-4,7-dihydro-5H-thieno[2,3-c]pyran-3,5-dicarboxylic acid 3-tert-butyl ester 5-ethyl ester (A) and 0.3 g of 2-amino-4,7-dihydro-5H-thieno[2,3-c]pyran-3,7-dicarboxylic acid 3-tert-butyl ester 7-ethyl ester (B) in 62 % combined yield. (A)1H-NMR (300 MHz, CDCl3) delta 5.96 (bs, 2H), 4.77-4.61 (m, 2H), 4.32-4.18 (m, 3H), 3.19-3.12 (m, 1H), 2.90-2.80 (m, 1H), 1.52 (s, 9H), 1.29 (t, 3H, J = 7 Hz).(B)1H-NMR (300 MHz, CDCl3) delta 5.10 (s, 1H), 4.28-4.13 (m, 3H), 3.98-3.91 (m, 1H), 2.82-2.76 (m, 2H), 1.51 (s, 9H), 1.31 (t, 3H, J = 7 Hz). |
|
With morpholine; sulphur; In ethanol; at 50℃; for 18h; |
To a solution of 4-oxo-tetrahydro-2H-pyran-2-carboxylic acid ethyl (0.6 g, 3.5 mmol) in absolute ethanol (6 ml) was added sulfur (0.12 g, 3.85 mmol) and tert-butyl cyanoacetate (0.64 g, 4.55 mmol). The solution was stirred under nitrogen in a 50 C. oil bath and morpholin (0.61 ml, 7.0 mmol) was added. The reaction was stirred for 18 hours and then cooled to ambient temperature and excess sulfur removed by filtration. The filtrate was concentrated in vacuo and reconstituted in ethyl acetate (50 ml). The organic phase was washed with brine (2*10 ml), dried (Na2SO4), filtered, and the solvent evaporated in vacuo. The residue was purified by silica gel chromatography using a gradient of ethyl acetate/hexane (20 to 25% gradient) as eluent. Pure fraction of the two isomers were collected and the solvent evaporated in vacuo which afforded 0.47 g of 2-amino-4,7-dihydro-5H-thieno[2,3-c]pyran-3,5-dicarboxylic acid 3-tert-butyl ester 5-ethyl ester (A) and 0.3 g of 2-amino-4,7-dihydro-5H-thieno[2,3-c]pyran-3,7-dicarboxylic acid 3-tert-butyl ester 7-ethyl ester (B) in 62% combined yield. (A)1H-NMR (300 MHz, CDCl3) delta 5.96 (bs, 2H), 4.77-4.61 (m, 2H), 4.32-4.18 (m, 3H), 3.19-3.12 (m, 1H), 2.90-2.80 (m, 1H), 1.52 (s, 9H), 1.29 (t, 3H, J=7 Hz).(B)1H-NMR (300 MHz, CDCl3) delta 5.10 (s, 1H), 4.28-4.13 (m, 3H), 3.98-3.91 (m, 1H), 2.82-2.76 (m, 2H), 1.51 (s, 9H), 1.31 (t, 3H, J=7 Hz). |
|
With morpholine; sulfur; In ethanol; at 50℃; for 18h; |
Example 30 2-(Oxalyl-amino)-5-phenylcarbamoyl-4,7-dihydro-5H-thieno[2,3-c]pyran-3 carboxylic acid; A solution of glyoxylic acid ethyl ester, polymer form (2.02 g, 8.9 mmol) and (3-methoxy-1-methylene-allyloxy)-trimethyl-silane (1.9 ml, 8.9 mmol, Danishefsky's diene) in benzene (12 ml) was placed under nitrogen. Zinc chloride (0.5N in tetrahydrofuran, 8.9 ml, 4.45 mmol) was added and the reaction stirred at ambient temperature for 72 h. The mixture was concentrated in vacuo, diluted with ethyl acetate (100 ml) and washed with 1 N hydrochloric acid (20 ml), saturated sodium bicarbonate (20 ml), and brine (20 ml). The organic layer was dried (Na2SO4), filtered, and the solvent evaporated in vacuo. The residue was purified by silica gel chromatography using a mixture of ethyl acetate/hexane (1:2) as eluant. Pure fractions were collected and the solvent evaporated in vacuo which afforded 1.2 g (75%) of 4-oxo-3,4-dihydro-2H-pyran-2-carboxylic acid ethyl ester as an oil.1H NMR (400 MHz, CDCl3) delta 7.40 (d, J=6, 1H), 5.48 (d, J=6, 1H), 5.01 (t, J=8, 1H), 4.28 (q, J=7, 2H), 2.85 (d, J=8, 2H), 1.29 (t, J=7, 3H).To a solution of the above of 4-oxo-3,4-dihydro-2H-pyran-2-carboxylic acid ethyl ester (1.0 g, 5.9 mmol) in ethyl acetate (12 ml) was added 10% palladium on activated carbon (0.15 g). The reaction was shaken on a Parr hydrogenator under a hydrogen atmosphere (30 psi) for 1.5 h. The mixture was filtered through celite and concentrated in vacuo. The residue was purified by silica gel chromatography sing diethyl ether as eluant. Pure fractions were collected and the solvent evaporated in vacuo which affording 0.6 g (60%) of 4-oxo-tetrahydro-2H-pyran-2-carboxylic acid ethyl as an oil.1H NMR (300 MHz, CDCl3) delta 4.41-4.35 (m, 1H), 4.26 (q, J=7, 2H), 3.81-3.70 (m, 1H), 2.73-2.58 (m, 3H), 2.44-2.36 (m, 1H), 1.29 (t, J=7, 3H).To a solution of 4-oxo-tetrahydro-2H-pyran-2-carboxylic acid ethyl (0.6 g, 3.5 mmol) in absolute ethanol (6 ml) was added sulfur (0.12 g, 3.85 mmol) and tert-butyl cyanoacetate (0.64 g, 4.55 mmol). The solution was stirred under nitrogen in a 50 C. oil bath and morpholin (0.61 ml, 7.0 mmol) was added. The reaction was stirred for 18 h. and then cooled to ambient temperature and excess sulfur removed by filtration. The filtrate was concentrated in vacuo and reconstituted in ethyl acetate (50 ml). The organic phase was washed with brine (2×10 ml), dried (Na2SO4), filtered, and the solvent evaporated in vacuo. The residue was purified by silica gel chromatography using a gradient of ethyl acetate/hexane (20 to 25% gradient) as eluant. Pure fraction of the two isomers were collected and the solvent evaporated in vacuo which afforded 0.47 g of 2-amino-4,7-dihydro-5H-thieno[2,3-c]pyran-3,5-dicarboxylic acid 3-tert-butyl ester 5-ethyl ester (A) and 0.3 g of 2-amino-4,7-dihydro-5H-thieno[2,3-c]pyran-3,7-dicarboxylic acid 3-tert-butyl ester 7-ethyl ester (B) in 62% combined yield.(A)1H NMR (300 MHz, CDCl3) delta 5.96 (bs, 2H), 4.77-4.61 (m, 2H), 4.32-4.18 (m, 3H), 3.19-3.12 (m, 1H), 2.90-2.80 (m, 1H), 1.52 (s, 9H), 1.29 (t, J=7, 3H).APCI-MS: [M+H]+=272.4 (loss of t-butyl)(B)1H NMR (300 MHz, CDCl3) delta5.10 (s, 1H), 4.28-4.13 (m, 3H), 3.98-3.91 (m, 1H), 2.82-2.76 (m, 2H), 1.51 (s, 9H), 1.31 (t, J=7, 3H).APCI-MS: [M+H]+=272.4 (loss of t-butyl)The above 2-amino-4,7-dihydro-5H-thieno[2,3-c]pyran-3,5-dicarboxylic acid 3-tert-butyl ester 5-ethyl ester (275 mg, 0.84 mmol) was dissolved in a mixture of ethanol (4 ml) and tetrahydrofuran (1 ml). Sodium hydroxide (1N, 1.6 ml, 1.68 mmol) was added and the reaction stirred at ambient temperature for 5 h. after which TLC analysis indicated that the reaction was complete. The reaction was monitored with a pH meter and neutralized with 1N hydrochloric acid until pH=6.9. The solution was concentrated in vacuo to give 2-amino-4+/-7-dihydro-5H-thieno[2,3-c]pyran-3,5-dicarboxylic acid 3-tert-butyl ester as a solid. Sodium chloride remained as an impurity.1H NMR (300 MHz, CD3OD) delta 4.67-4.54 (m, 2H), 4.00-3.95 (m, 1H), 3.20-3.12 (m, 1H), 2.74-2.63 (m, 1H), 1.54 (s, 9H).APCI-MS: [M+H]+=300.0To a solution of the above 2-amino-4,7-dihydro-5H-thieno[2,3-c]pyran-3,5-dicarboxylic acid 3-tert-butyl ester (94 mg, 0.31 mmol) and 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride (72 mg, 0.37 mmol) in distilled dichloromethane (4 ml) under nitrogen was added aniline (32 mul, 0.34 mmol) followed by 2,6-lutidine (0.11 ml, 0.93 mmol). The reaction was stirred for 72 h., concentrated in vacuo and reconstituted in ethyl acetate (30 ml). The organic layer was washed with 1% hydrochloric acid (10 ml), saturated sodium bicarbonate (10 ml), brine (10 ml), dried (Na2SO4), filtered, and the solvent evaporated in vacuo to give 51 mg (45%) of 2-amino-5-phenylcarbamoyl-4,7-dihydro-5H-thieno[2,3-c]pyran-3-carboxylic acid tert-butyl ester as a solid.1H NMR (400 MHz, CDCl3) delta 8.40 (s, 1H), 7.60 (d, 1H, J=7), 7.49 (d, 1H, =8), 7.34 (t, 1H, J=8), 7.32 (t, 1H, J=8), 7.13 (t, 1H, J=7)... |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 120K+ Compounds
120K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping