| Identification | Back Directory | [Name]
1-BUTEN-3-YNE | [CAS]
689-97-4 | [Synonyms]
Butenyne
1-Butenyne
Buten-3-yne
1-BUTEN-3-YNE
1-Butyn-3-ene
ethynyl-ethen
Ethynylethene
3-BUTEN-1-YNE
But-1-en-3-yne
CH2=CHCequivCH
VINYLACETYLENE
1-Butene-3-yne
Buten-3-yne, 1-
Ethene, ethynyl-
Monovinylacetylene
1-Buten-3-yne (8CI, 9CI)
3-BUTEN-1-YNE 50% IN XYLENE
1-BUTEN-3-YNE, 50% in XYLENE
1-buten-3-yne,vinylacetylene
1-BUTEN-3-YNE ISO 9001:2015 REACH | [EINECS(EC#)]
211-713-3 | [Molecular Formula]
C4H4 | [MDL Number]
MFCD00053683 | [MOL File]
689-97-4.mol | [Molecular Weight]
52.07 |
| Hazard Information | Back Directory | [Description]
Vinylacetylene, also known as butenyne, monovinylacetylene, 1-butene-3-yne, 1-butyn-
3-ene, 3-buten-1-yne, buten-3-yne, ethynylethene, 1-butenyne and vinylethyne, is a
derivative of acetylene. | [Chemical Properties]
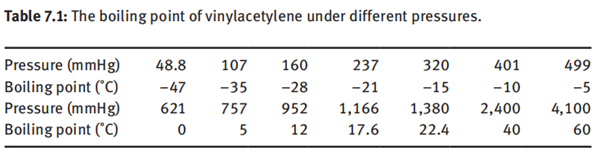
Vinylacetylene is a gas with a smell similar to acetylene. The melting point is –118
to –120°C, the boiling point is 5.5 °C under atmospheric pressure, and the boiling
point of vinylacetylene under different pressures is shown in Table 7.1.
 | [Uses]
Vinylacetylene is mainly used for the production of chloroprene, leaf alcohol and
methanol adhesives. It is also used for the production of divinyl ether and methyl
vinyl (methyl) ketone-based polymer. | [Definition]
ChEBI: Butenyne is an acyclic acetylene, an acyclic olefin, an enyne and a terminal acetylenic compound. | [General Description]
A colorless gas or liquid. Derived by the dimerization of acetylene. Used in the synthesis of neoprene and for other organic syntheses. | [Air & Water Reactions]
Highly flammable. Oxidizes in air to form unstable peroxides that may explode spontaneously. | [Reactivity Profile]
1-BUTEN-3-YNE(689-97-4) may react vigorously with strong oxidizing agents. May react exothermically with reducing agents In the presence of various catalysts (such as acids) or initiators, may undergo exothermic addition polymerization reactions. Forms an explosive salt with silver nitrate. Thermally unstable, under pressure, in combination with 1,3-butadiene [Loss Prev., 1971, 5, 67]. | [Synthesis]
Acetylene is dimerized in the presence of hydrochloric acid solution of cuprous chloride and ammonium chloride to obtain vinylacetylene. |
|
| Company Name: |
CHEMSTEP
|
| Tel: |
33 (0) 556 405 186 |
| Website: |
www.chemstep.com |
|