| Identification | Back Directory | [Name]
erythro-Glycopyrronium bromide | [CAS]
51186-83-5 | [Synonyms]
GlycopyrroniuM BroMide API
erythro-Glycopyrronium bromide
Glycopyrronium bromide
(CHF 5259
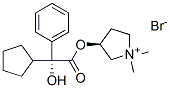
(R*,S*)-3-[(Cyclopentylhydroxyphenylacetyl)oxy]-1,1-dimethylpyrrolidinium bromide
Pyrrolidinium, 3-[(cyclopentylhydroxyphenylacetyl)oxy]-1,1-dimethyl-, bromide, (R*,S*)-
(3S)-3-{[(2R)-2-Cyclopentyl-2-hydroxy-2-phenylacetyl]oxy}-1,1-dimethylpyrrolidinium bromide
Pyrrolidinium, 3-[(cyclopentylhydroxyphenylacetyl)oxy]-1,1-dimethyl-, bromide, (R*,S*)-(+-)- | [EINECS(EC#)]
209-887-0 | [Molecular Formula]
C19H28NO3.Br | [MDL Number]
MFCD31381925 | [MOL File]
51186-83-5.mol |
| Chemical Properties | Back Directory | [Appearance]
White or almost white, crystalline powder. | [Melting point ]
175℃ | [solubility ]
Freely soluble in water, soluble in ethanol (96 per cent), very slightly soluble in methylene chloride. | [InChI]
InChI=1/C19H28NO3.BrH/c1-20(2)13-12-17(14-20)23-18(21)19(22,16-10-6-7-11-16)15-8-4-3-5-9-15;/h3-5,8-9,16-17,22H,6-7,10-14H2,1-2H3;1H/q+1;/p-1/t17-,19-;/s3 | [InChIKey]
VPNYRYCIDCJBOM-SCHKGOGONA-M | [SMILES]
[N+]1(C)(C)CC[C@H](OC([C@](C2CCCC2)(O)C2=CC=CC=C2)=O)C1.[Br-] |&1:5,8,r| | [Uses]
Anticholinergic |
| Hazard Information | Back Directory | [Chemical Properties]
White or almost white, crystalline powder. | [Description]
Glycopyrronium bromide or NVA 237 is a once-daily inhaled antimuscarinic bronchodilator newly approved by the European Commission for use in stable COPD. It is characterized by a quaternary ammonium structure, which reduces its oral bioavailability and, consequently, the systematic effects of any swallowed portion of the drug.
Animal studies suggest that there are fewer cardiovascular effects compared to tiotropium because of the higher affinity to M3 versus M2 muscarinic receptors. Human studies showed fast onset of action, sustained bronchodilatation for 24 h, reduced dynamic hyperinflation of the lungs, and improved exercise tolerance. Glycopyrronium bromide in COPD airways clinical trial series (GLOW1–3) are the major clinical trials studying the efficacy and safety of glycopyrronium[1].
| [Definition]
ChEBI: (2R,3S)-glycopyrronium bromide is a glycopyrronium bromide that has (2R,3S)-configuration. The racemate, ritropirronium bromide, is used for treatment of chronic obstructive pulmonary disease. It is an enantiomer of a (2S,3R)-glycopyrronium bromide. | [Indications]
Glycopyrronium bromide is indicated for use as a preoperative antimuscarinic to reduce salivary, tracheobronchial, and pharyngeal secretions; to reduce the volume and free acidity of gastric secretions, and to block cardiac vagal inhibitory reflexes during induction of anaesthesia and intubation when indicated. Glycopyrronium bromide may be used intraoperatively to counteract drug-induced or vagal traction reflexes with the associated arrhythmias. Glycopyrronium bromide protects against the peripheral muscarinic effects (e.g., bradycardia and excessive secretions) of cholinergic agents such as neostigmine and pyridostigmine given to reverse the neuromuscular blockade due to non-depolarising muscle relaxants.
| [Side effects]
The following reported adverse reactions are extensions of glycopyrronium bromide’s fundamentalpharmacological actions:
Cardiovascular: Tachycardia, ventricular fibrillation, bradycardia, palpitation and arrhythmia, hypertension, hypotension, cardiac arrest, heart block, prolonged QTc interval.
Dermatological: Flushing and inhibition of sweating. Severe allergic reactions or drug idiosyncrasies including urticaria and other dermal manifestations, pruritus, dry skin.
Gastrointestinal: Nausea, vomiting, dry mouth, constipation, taste alterations, including loss of taste.
Genitourinary: Urinary hesitancy and retention, impotence.
Ocular: Blurred vision due to mydriasis, cycloplegia, photophobia, increased ocular tension.
Nervous System: Inhibition of transmission at neuromuscular junction, headache, nervousness, drowsiness, dizziness, seizure, insomnia, some degree of mental confusion, especially in the elderly, hyperexcitability in children.
Pregnancy and perinatal: Suppression of lactation.
Respiratory System: Respiratory arrest.
General: Hyperpyrexia, bloated feeling, anaphylaxis/anaphylactoid reaction, hypersensitivity. Injection site reactions including pruritus, oedema, erythema, pain have been reported rarely.
| [References]
[1] Alexander G. Mathioudakis, Victoria Chatzimavridou-Grigoriadou, Georgios A. Mathioudakis.“Chapter 16 - Drugs that act on the respiratory tract.” 2014: 241-256. |
|
|